Inhibition of leptin regulation of parasympathetic signaling as a cause of extreme body weight-associated asthma
- PMID: 23312282
- PMCID: PMC3815545
- DOI: 10.1016/j.cmet.2012.12.004
Inhibition of leptin regulation of parasympathetic signaling as a cause of extreme body weight-associated asthma
Erratum in
- Cell Metab. 2013 Mar 5;17(3):463-4
Abstract
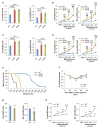
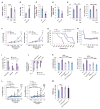
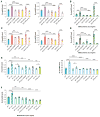
Impaired lung function caused by decreased airway diameter (bronchoconstriction) is frequently observed whether body weight is abnormally high or low. That these opposite conditions affect the airways similarly suggests that the regulation of airway diameter and body weight are intertwined. We show here that, independently of its regulation of appetite, melanocortin pathway, or sympathetic tone, leptin is necessary and sufficient to increase airway diameter by signaling through its cognate receptor in cholinergic neurons. The latter decreases parasympathetic signaling through the M(3) muscarinic receptor in airway smooth muscle cells, thereby increasing airway diameter without affecting local inflammation. Accordingly, decreasing parasympathetic tone genetically or pharmacologically corrects bronchoconstriction and normalizes lung function in obese mice regardless of bronchial inflammation. This study reveals an adipocyte-dependent regulation of bronchial diameter whose disruption contributes to the impaired lung function caused by abnormal body weight. These findings may be of use in the management of obesity-associated asthma.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures
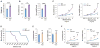



Similar articles
-
Hyperinsulinemia potentiates airway responsiveness to parasympathetic nerve stimulation in obese rats.Am J Respir Cell Mol Biol. 2014 Aug;51(2):251-61. doi: 10.1165/rcmb.2013-0452OC. Am J Respir Cell Mol Biol. 2014. PMID: 24605871 Free PMC article.
-
Muscarinic receptor-mediated bronchoconstriction is coupled to caveolae in murine airways.Am J Physiol Lung Cell Mol Physiol. 2010 May;298(5):L626-36. doi: 10.1152/ajplung.00261.2009. Epub 2009 Dec 18. Am J Physiol Lung Cell Mol Physiol. 2010. PMID: 20023174 Free PMC article.
-
Muscarinic acetylcholine receptors and airway diseases.Pharmacol Ther. 2003 Apr;98(1):59-69. doi: 10.1016/s0163-7258(03)00004-4. Pharmacol Ther. 2003. PMID: 12667888 Review.
-
Beta2-agonist and anticholinergic drugs in the treatment of lung disease.Proc Am Thorac Soc. 2005;2(4):305-10; discussion 311-2. doi: 10.1513/pats.200504-038SR. Proc Am Thorac Soc. 2005. PMID: 16267353 Review.
-
Acetylcholine beyond bronchoconstriction: roles in inflammation and remodeling.Trends Pharmacol Sci. 2015 Mar;36(3):164-71. doi: 10.1016/j.tips.2014.11.005. Epub 2014 Dec 13. Trends Pharmacol Sci. 2015. PMID: 25511176 Review.
Cited by
-
Obesity-associated Airway Hyperresponsiveness: Mechanisms Underlying Inflammatory Markers and Possible Pharmacological Interventions.Endocr Metab Immune Disord Drug Targets. 2024;24(9):1053-1068. doi: 10.2174/0118715303256440231028072049. Endocr Metab Immune Disord Drug Targets. 2024. PMID: 37957906 Review.
-
Inhibiting the Notch signaling pathway suppresses Th17-associated airway hyperresponsiveness in obese asthmatic mice.Lab Invest. 2019 Dec;99(12):1784-1794. doi: 10.1038/s41374-019-0294-x. Epub 2019 Aug 13. Lab Invest. 2019. PMID: 31409887
-
BMI but not central obesity predisposes to airway closure during bronchoconstriction.Respirology. 2019 Jun;24(6):543-550. doi: 10.1111/resp.13478. Epub 2019 Jan 29. Respirology. 2019. PMID: 30694011 Free PMC article.
-
A pilot randomized controlled trial of pioglitazone for the treatment of poorly controlled asthma in obesity.Respir Res. 2015 Nov 26;16:143. doi: 10.1186/s12931-015-0303-6. Respir Res. 2015. PMID: 26610598 Free PMC article. Clinical Trial.
-
Control of respiratory and cardiovascular functions by leptin.Life Sci. 2015 Mar 15;125:25-31. doi: 10.1016/j.lfs.2015.01.019. Epub 2015 Jan 30. Life Sci. 2015. PMID: 25645056 Free PMC article. Review.
References
-
- Barnes PJ. The pharmacological properties of tiotropium. Chest. 2000;117(2, Suppl):63S–66S. - PubMed
-
- Butler AA, Kesterson RA, Khong K, Cullen MJ, Pelleymounter MA, Dekoning J, Baetscher M, Cone RD. A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology. 2000;141:3518–3521. - PubMed
-
- Canning BJ. Reflex regulation of airway smooth muscle tone. J Appl Physiol. 2006;101:971–985. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

