Adoptive transfer of Tc1 or Tc17 cells elicits antitumor immunity against established melanoma through distinct mechanisms
- PMID: 23315072
- PMCID: PMC3563723
- DOI: 10.4049/jimmunol.1201989
Adoptive transfer of Tc1 or Tc17 cells elicits antitumor immunity against established melanoma through distinct mechanisms
Abstract
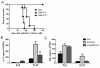
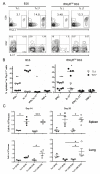
Adoptive cell transfer (ACT) of ex vivo-activated autologous tumor-reactive T cells is currently one of the most promising approaches for cancer immunotherapy. Recent studies provided some evidence that IL-17-producing CD8(+) (Tc17) cells may exhibit potent antitumor activity, but the specific mechanisms have not been completely defined. In this study, we used a murine melanoma lung-metastasis model and tested the therapeutic effects of gp100-specific polarized type I CD8(+) cytotoxic T (Tc1) or Tc17 cells combined with autologous bone marrow transplantation after total body irradiation. Bone marrow transplantation combined with ACT of antitumor (gp100-specific) Tc17 cells significantly suppressed the growth of established melanoma, whereas Tc1 cells induced long-term tumor regression. After ACT, Tc1 cells maintained their phenotype to produce IFN-γ, but not IL-17. However, although Tc17 cells largely preserved their ability to produce IL-17, a subset secreted IFN-γ or both IFN-γ and IL-17, indicating the plasticity of Tc17 cells in vivo. Furthermore, after ACT, the Tc17 cells had a long-lived effector T cell phenotype (CD127(hi)/KLRG-1(low)) as compared with Tc1 cells. Mechanistically, Tc1 cells mediated antitumor immunity primarily through the direct effect of IFN-γ on tumor cells. In contrast, despite the fact that some Tc17 cells also secreted IFN-γ, Tc17-mediated antitumor immunity was independent of the direct effects of IFN-γ on the tumor. Nevertheless, IFN-γ played a critical role by creating a microenvironment that promoted Tc17-mediated antitumor activity. Taken together, these studies demonstrate that both Tc1 and Tc17 cells can mediate effective antitumor immunity through distinct effector mechanisms, but Tc1 cells are superior to Tc17 cells in mediating tumor regression.
Figures



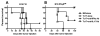
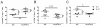
Similar articles
-
Type 1 and type 2 CD8+ effector T cell subpopulations promote long-term tumor immunity and protection to progressively growing tumor.J Immunol. 2000 Jan 15;164(2):916-25. doi: 10.4049/jimmunol.164.2.916. J Immunol. 2000. PMID: 10623840
-
Adoptive transfer of tumor-specific Tc17 effector T cells controls the growth of B16 melanoma in mice.J Immunol. 2010 Apr 15;184(8):4215-27. doi: 10.4049/jimmunol.0902995. Epub 2010 Mar 17. J Immunol. 2010. PMID: 20237297 Free PMC article.
-
Dendritic Cells in Irradiated Mice Trigger the Functional Plasticity and Antitumor Activity of Adoptively Transferred Tc17 Cells via IL12 Signaling.Clin Cancer Res. 2015 Jun 1;21(11):2546-57. doi: 10.1158/1078-0432.CCR-14-2294. Epub 2015 Apr 22. Clin Cancer Res. 2015. PMID: 25904754 Free PMC article.
-
Tc17 Cells in Immunity and Systemic Autoimmunity.Int Rev Immunol. 2015;34(4):318-31. doi: 10.3109/08830185.2014.954698. Epub 2014 Sep 26. Int Rev Immunol. 2015. PMID: 25259411 Review.
-
New Insights on CD8+ T Cells in Inflammatory Bowel Disease and Therapeutic Approaches.Front Immunol. 2021 Oct 11;12:738762. doi: 10.3389/fimmu.2021.738762. eCollection 2021. Front Immunol. 2021. PMID: 34707610 Free PMC article. Review.
Cited by
-
The inducible costimulator augments Tc17 cell responses to self and tumor tissue.J Immunol. 2015 Feb 15;194(4):1737-47. doi: 10.4049/jimmunol.1401082. Epub 2015 Jan 9. J Immunol. 2015. PMID: 25576595 Free PMC article.
-
Activation of CD1d-restricted natural killer T cells can inhibit cancer cell proliferation during chemotherapy by promoting the immune responses in murine mesothelioma.Cancer Immunol Immunother. 2014 Dec;63(12):1285-96. doi: 10.1007/s00262-014-1597-9. Epub 2014 Sep 3. Cancer Immunol Immunother. 2014. PMID: 25183171 Free PMC article.
-
IL-27 Receptor Signaling on T cells Augments GVHD Severity through Enhancing Th1 Responses.J Immunol Res Ther. 2018;3(1):151-157. Epub 2018 Jun 7. J Immunol Res Ther. 2018. PMID: 30906912 Free PMC article.
-
CD38-NAD+Axis Regulates Immunotherapeutic Anti-Tumor T Cell Response.Cell Metab. 2018 Jan 9;27(1):85-100.e8. doi: 10.1016/j.cmet.2017.10.006. Epub 2017 Nov 9. Cell Metab. 2018. PMID: 29129787 Free PMC article.
-
PD-1 limits differentiation and plasticity of Tc17 cells.Front Immunol. 2023 Apr 28;14:1104730. doi: 10.3389/fimmu.2023.1104730. eCollection 2023. Front Immunol. 2023. PMID: 37205114 Free PMC article.
References
-
- Carter LL, Dutton RW. Type 1 and type 2: a fundamental dichotomy for all T-cell subsets. Curr Opin Immunol. 1996;8:336–342. - PubMed
-
- Sad S, Marcotte R, Mosmann TR. Cytokine-induced differentiation of precursor mouse CD8+ T cells into cytotoxic CD8+ T cells secreting Th1 or Th2 cytokines. Immunity. 1995;2:271–279. - PubMed
-
- Muranski P, Boni A, Antony PA, Cassard L, Irvine KR, Kaiser A, Paulos CM, Palmer DC, Touloukian CE, Ptak K, Gattinoni L, Wrzesinski C, Hinrichs CS, Kerstann KW, Feigenbaum L, Chan C-C, Restifo NP. Tumor-specific Th17-polarized cells eradicate large established melanoma. Blood. 2008;112:362–373. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

