Role of β-catenin-regulated CCN matricellular proteins in epithelial repair after inflammatory lung injury
- PMID: 23316072
- PMCID: PMC3602744
- DOI: 10.1152/ajplung.00180.2012
Role of β-catenin-regulated CCN matricellular proteins in epithelial repair after inflammatory lung injury
Abstract
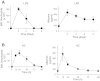
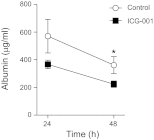
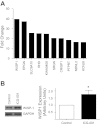
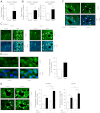
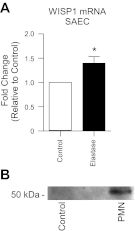
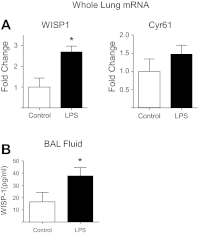
Repair of the lung epithelium after injury is integral to the pathogenesis and outcomes of diverse inflammatory lung diseases. We previously reported that β-catenin signaling promotes epithelial repair after inflammatory injury, but the β-catenin target genes that mediate this effect are unknown. Herein, we examined which β-catenin transcriptional coactivators and target genes promote epithelial repair after inflammatory injury. Transmigration of human neutrophils across cultured monolayers of human lung epithelial cells resulted in a fall in transepithelial resistance and the formation of discrete areas of epithelial denudation ("microinjury"), which repaired via cell spreading by 96 h. In mice treated with intratracheal (i.t.) LPS or keratinocyte chemokine, neutrophil emigration was associated with increased permeability of the lung epithelium, as determined by increased bronchoalveolar lavage (BAL) fluid albumin concentration, which decreased over 3-6 days. Activation of β-catenin/p300-dependent gene expression using the compound ICG-001 accelerated epithelial repair in vitro and in murine models. Neutrophil transmigration induced epithelial expression of the β-catenin/p300 target genes Wnt-induced secreted protein (WISP) 1 and cysteine-rich (Cyr) 61, as determined by real-time PCR (qPCR) and immunostaining. Purified neutrophil elastase induced WISP1 upregulation in lung epithelial cells, as determined by qPCR. WISP1 expression increased in murine lungs after i.t. LPS, as determined by ELISA of the BAL fluid and qPCR of whole lung extracts. Finally, recombinant WISP1 and Cyr61 accelerated repair, and Cyr61-neutralizing antibodies delayed repair of the injured epithelium in vitro. We conclude that β-catenin/p300-dependent expression of WISP1 and Cyr61 is critical for epithelial repair and represents a potential therapeutic target to promote epithelial repair after inflammatory injury.
Figures


Comment in
-
β-Catenin and CCNs in lung epithelial repair.Am J Physiol Lung Cell Mol Physiol. 2013 May 1;304(9):L579-81. doi: 10.1152/ajplung.00073.2013. Epub 2013 Mar 22. Am J Physiol Lung Cell Mol Physiol. 2013. PMID: 23525784 Free PMC article. No abstract available.
References
-
- Aarbiou J, Ertmann M, van Wetering S, van Noort P, Rook D, Rabe KF, Litvinov SV, van Krieken JH, de Boer WI, Hiemstra PS. Human neutrophil defensins induce lung epithelial cell proliferation in vitro. J Leukoc Biol 72: 167–174, 2002 - PubMed
-
- Adamson A, Perkins S, Brambilla E, Tripp S, Holden J, Travis W, Guinee D., Jr Proliferation, C-myc, and cyclin D1 expression in diffuse alveolar damage: potential roles in pathogenesis and implications for prognosis. Hum Pathol 30: 1050–1057, 1999 - PubMed
-
- Adamson IY, Bowden DH. The type 2 cell as progenitor of alveolar epithelial regeneration. A cytodynamic study in mice after exposure to oxygen. Lab Invest 30: 35–42, 1974 - PubMed
-
- Ashino Y, Ying X, Dobbs LG, Bhattacharya J. [Ca(2+)](i) oscillations regulate type II cell exocytosis in the pulmonary alveolus. Am J Physiol Lung Cell Mol Physiol 279: L5–L13, 2000 - PubMed
-
- Atabai K, Ishigaki M, Geiser T, Ueki I, Matthay MA, Ware LB. Keratinocyte growth factor can enhance alveolar epithelial repair by nonmitogenic mechanisms. Am J Physiol Lung Cell Mol Physiol 283: L163–L169, 2002 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

