Developmental regulation of spatio-temporal patterns of cortical circuit activation
- PMID: 23316135
- PMCID: PMC3539829
- DOI: 10.3389/fncel.2012.00065
Developmental regulation of spatio-temporal patterns of cortical circuit activation
Abstract
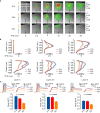
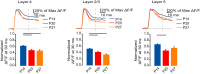
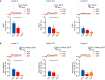
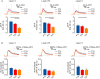
Neural circuits are refined in an experience-dependent manner during early postnatal development. How development modulates the spatio-temporal propagation of activity through cortical circuits is poorly understood. Here we use voltage-sensitive dye imaging (VSD) to show that there are significant changes in the spatio-temporal patterns of intracortical signals in primary visual cortex (V1) from postnatal day 13 (P13), eye opening, to P28, the peak of the critical period for rodent visual cortical plasticity. Upon direct stimulation of layer 4 (L4), activity spreads to L2/3 and to L5 at all ages. However, while from eye opening to the peak of the critical period, the amplitude and persistence of the voltage signal decrease, peak activation is reached more quickly and the interlaminar gain increases with age. The lateral spread of activation within layers remains unchanged throughout the time window under analysis. These developmental changes in spatio-temporal patterns of intracortical circuit activation are mediated by differences in the contributions of excitatory and inhibitory synaptic components. Our results demonstrate that after eye opening the circuit in V1 is refined through a progression of changes that shape the spatio-temporal patterns of circuit activation. Signals become more efficiently propagated across layers through developmentally regulated changes in interlaminar gain.
Keywords: AMPA; GABA; NMDA; microcircuitry; postnatal development; signal propagation; synaptic plasticity; visual cortex.
Figures



References
-
- Blue M. E., Parnavelas J. G. (1983). The formation and maturation of synapses in the visual cortex of the rat. II. Quantitative analysis. J. Neurocytol. 12, 697–712 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

