Sulfated pentagalloylglucoside is a potent, allosteric, and selective inhibitor of factor XIa
- PMID: 23316863
- PMCID: PMC3574227
- DOI: 10.1021/jm301338q
Sulfated pentagalloylglucoside is a potent, allosteric, and selective inhibitor of factor XIa
Abstract
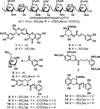
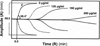
Inhibition of factor XIa (FXIa) is a novel paradigm for developing anticoagulants without major bleeding consequences. We present the discovery of sulfated pentagalloylglucoside (6) as a highly selective inhibitor of human FXIa. Biochemical screening of a focused library led to the identification of 6, a sulfated aromatic mimetic of heparin. Inhibitor 6 displayed a potency of 551 nM against FXIa, which was at least 200-fold more selective than other relevant enzymes. It also prevented activation of factor IX and prolonged human plasma and whole blood clotting. Inhibitor 6 reduced V(MAX) of FXIa hydrolysis of chromogenic substrate without affecting the K(M), suggesting an allosteric mechanism. Competitive studies showed that 6 bound in the heparin-binding site of FXIa. No allosteric small molecule has been discovered to date that exhibits equivalent potency against FXIa. Inhibitor 6 is expected to open up a major route to allosteric FXIa anticoagulants with clinical relevance.
Figures






References
-
- Henry BL, Desai UR. Anticoagulants: Drug discovery and development. In: Rotella D, Abraham DJ, editors. Burger’s Medicinal Chemistry. 7th ed. New York: John Wiley and Sons; 2010. pp. 365–408.
-
- Bates SM, Weitz JI. The status of new anticoagulants. Br. J. Haematol. 2006;134:3–19. - PubMed
-
- Hirsh J, Anand SS, Halperin JL, Fuster V. Guide to anticoagulant therapy: Heparin: a statement for healthcare professionals from the American Heart Association. Circulation. 2001;103:2994–3018. - PubMed
-
- Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians. Evidence-based clinical practice guidelines, 8th ed. Chest. 2008;133:160S–198S. - PubMed
-
- Eikelboom JW, Wallentin L, Connolly SJ, Ezekowitz M, Healey JS, Oldgren J, Yang S, Alings M, Kaatz S, Hohnloser SH, Diener H-C, Franzosi MG, Huber K, Reilly P, Varrone J, Yusuf S. Risk of Bleeding With 2 Doses of Dabigatran Compared With Warfarin in Older and Younger Patients With Atrial Fibrillation. An analysis of the randomized evaluation of long-term anticoagulant therapy (RE-LY) Trial. Circulation. 2011;123:2363–2372. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information

