Prostaglandin E₂-induced intercellular adhesion molecule-1 expression is mediated by cAMP/Epac signalling modules in bEnd.3 brain endothelial cells
- PMID: 23317035
- PMCID: PMC3682708
- DOI: 10.1111/bph.12103
Prostaglandin E₂-induced intercellular adhesion molecule-1 expression is mediated by cAMP/Epac signalling modules in bEnd.3 brain endothelial cells
Abstract
Background and purpose: Prostaglandin E₂ (PGE₂) has been implicated in the regulation of adhesion molecules, leukocyte adhesion and infiltration into inflamed site. However, the underlying mechanism therein involved remains ill-defined. In this study, we explored its cellular mechanism of action in the regulation of the intercellular adhesion molecule-1 (ICAM-1) expression in the brain endothelial cells.
Experimental approach: bEnd.3 cells, the murine cerebrovascular endothelial cell line and primary mouse brain endothelial cells were treated with PGE₂ with or without agonists/antagonists of PGE₂ receptors and associated signalling molecules. ICAM-1 expression, Akt phosphorylation and activity of NF-κB were determined by reverse transcription polymerase chain reaction (RT-PCR), immunoblot analysis, luciferase assay and immunocytochemistry.
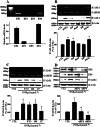
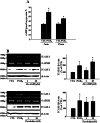
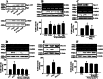
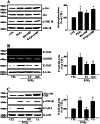
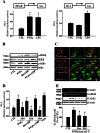
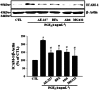
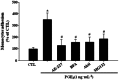
Key results: PGE₂ significantly up-regulated the expression of ICAM-1, which was blocked by EP4 antagonist (ONO-AE2-227) and knock-down of EP4. PGE₂ effects were mimicked by forskolin, dibutyryl cAMP (dbcAMP) and an exchange protein directly activated by cAMP (Epac) activator (8-Cpt-cAMP) but not a protein kinase A activator (N⁶-Bnz-cAMP). PGE₂-induced ICAM-1 expression was reduced by knock-down of Epac1. A PI3K specific inhibitor (LY294002), Akt inhibitor VIII (Akti) and NF-κB inhibitors (Bay-11-7082 and MG-132) attenuated the induction of ICAM-1 by PGE₂. PGE₂, dbcAMP and 8-Cpt-cAMP induced the phosphorylation of Akt, IκB kinase and IκBα and the translocation of p65 to the nucleus and increased NF-κB dependent reporter gene activity, which was diminished by Akti.
Conclusion and implications: Our findings suggest that PGE₂ induces ICAM-1 expression via EP4 receptor and Epac/Akt/NF-κB signalling pathway in bEnd.3 brain endothelial cells, supporting its pathophysiological role in brain inflammation.
© 2013 The Authors. British Journal of Pharmacology © 2013 The British Pharmacological Society.
Figures

References
-
- Aktan S, Aykut C, Oktay S, Yegen B, Keles E, Aykac I, et al. The alterations of leukotriene C4 and prostaglandin E2 levels following different ischemic periods in rat brain tissue. Prostaglandins Leukot Essent Fatty Acids. 1991;42:67–71. - PubMed
-
- Aloisi F, De Simone R, Columba-Cabezas S, Levi G. Opposite effects of interferon-gamma and prostaglandin E2 on tumor necrosis factor and interleukin-10 production in microglial: a regulatory loop controlling microglia pro- and anti-inflammatory activities. J Neurosci Res. 1999;56:571–580. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

