High yield production of myristoylated Arf6 small GTPase by recombinant N-myristoyl transferase
- PMID: 23319116
- PMCID: PMC3620099
- DOI: 10.4161/sgtp.22895
High yield production of myristoylated Arf6 small GTPase by recombinant N-myristoyl transferase
Abstract
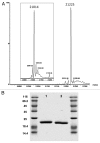
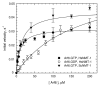
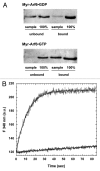
Small GTP-binding proteins of the Arf family (Arf GTPases) interact with multiple cellular partners and with membranes to regulate intracellular traffic and organelle structure. Understanding the underlying molecular mechanisms requires in vitro biochemical assays to test for regulations and functions. Such assays should use proteins in their cellular form, which carry a myristoyl lipid attached in N-terminus. N-myristoylation of recombinant Arf GTPases can be achieved by co-expression in E. coli with a eukaryotic N-myristoyl transferase. However, purifying myristoylated Arf GTPases is difficult and has a poor overall yield. Here we show that human Arf6 can be N-myristoylated in vitro by recombinant N-myristoyl transferases from different eukaryotic species. The catalytic efficiency depended strongly on the guanine nucleotide state and was highest for Arf6-GTP. Large-scale production of highly pure N-myristoylated Arf6 could be achieved, which was fully functional for liposome-binding and EFA6-stimulated nucleotide exchange assays. This establishes in vitro myristoylation as a novel and simple method that could be used to produce other myristoylated Arf and Arf-like GTPases for biochemical assays.
Keywords: Arf; Arf-like; myristoylation; small GTPases.
Figures
Similar articles
-
EFA6 controls Arf1 and Arf6 activation through a negative feedback loop.Proc Natl Acad Sci U S A. 2014 Aug 26;111(34):12378-83. doi: 10.1073/pnas.1409832111. Epub 2014 Aug 11. Proc Natl Acad Sci U S A. 2014. PMID: 25114232 Free PMC article.
-
Preparation of myristoylated Arf1 and Arf6.Methods Enzymol. 2005;404:164-74. doi: 10.1016/S0076-6879(05)04016-4. Methods Enzymol. 2005. PMID: 16413267
-
NMR structural studies of the myristoylated N-terminus of ADP ribosylation factor 6 (Arf6).FEBS Lett. 2006 Jul 24;580(17):4296-301. doi: 10.1016/j.febslet.2006.06.086. Epub 2006 Jul 7. FEBS Lett. 2006. PMID: 16839550
-
The role of ADP-ribosylation factor and SAR1 in vesicular trafficking in plants.Biochim Biophys Acta. 2004 Jul 1;1664(1):9-30. doi: 10.1016/j.bbamem.2004.04.005. Biochim Biophys Acta. 2004. PMID: 15238254 Review.
-
Allosteric regulation of Arf GTPases and their GEFs at the membrane interface.Small GTPases. 2016 Oct;7(4):283-296. doi: 10.1080/21541248.2016.1215778. Epub 2016 Jul 22. Small GTPases. 2016. PMID: 27449855 Free PMC article. Review.
Cited by
-
Binding with heat shock cognate protein HSC70 fine-tunes the Golgi association of the small GTPase ARL5B.J Biol Chem. 2021 Dec;297(6):101422. doi: 10.1016/j.jbc.2021.101422. Epub 2021 Nov 16. J Biol Chem. 2021. PMID: 34798070 Free PMC article.
-
Chlamydia Hijacks ARF GTPases To Coordinate Microtubule Posttranslational Modifications and Golgi Complex Positioning.mBio. 2017 May 2;8(3):e02280-16. doi: 10.1128/mBio.02280-16. mBio. 2017. PMID: 28465429 Free PMC article.
-
EFA6 controls Arf1 and Arf6 activation through a negative feedback loop.Proc Natl Acad Sci U S A. 2014 Aug 26;111(34):12378-83. doi: 10.1073/pnas.1409832111. Epub 2014 Aug 11. Proc Natl Acad Sci U S A. 2014. PMID: 25114232 Free PMC article.
-
Integrated conformational and lipid-sensing regulation of endosomal ArfGEF BRAG2.PLoS Biol. 2013 Sep;11(9):e1001652. doi: 10.1371/journal.pbio.1001652. Epub 2013 Sep 10. PLoS Biol. 2013. PMID: 24058294 Free PMC article.
-
In vitro reconstitution reveals phosphoinositides as cargo-release factors and activators of the ARF6 GAP ADAP1.Proc Natl Acad Sci U S A. 2021 Jan 5;118(1):e2010054118. doi: 10.1073/pnas.2010054118. Epub 2020 Dec 18. Proc Natl Acad Sci U S A. 2021. PMID: 33443153 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
