Salmonella polarises peptide-MHC-II presentation towards an unconventional Type B CD4+ T-cell response
- PMID: 23319341
- PMCID: PMC3816330
- DOI: 10.1002/eji.201242983
Salmonella polarises peptide-MHC-II presentation towards an unconventional Type B CD4+ T-cell response
Abstract
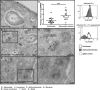
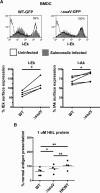
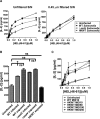
Distinct peptide-MHC-II complexes, recognised by Type A and B CD4(+) T-cell subsets, are generated when antigen is loaded in different intracellular compartments. Conventional Type A T cells recognize their peptide epitope regardless of the route of processing, whereas unconventional Type B T cells only recognise exogenously supplied peptide. Type B T cells are implicated in autoimmune conditions and may break tolerance by escaping negative selection. Here we show that Salmonella differentially influences presentation of antigen to Type A and B T cells. Infection of bone marrow-derived dendritic cells (BMDCs) with Salmonella enterica serovar Typhimurium (S. Typhimurium) reduced presentation of antigen to Type A T cells but enhanced presentation of exogenous peptide to Type B T cells. Exposure to S. Typhimurium was sufficient to enhance Type B T-cell activation. Salmonella Typhimurium infection reduced surface expression of MHC-II, by an invariant chain-independent trafficking mechanism, resulting in accumulation of MHC-II in multi-vesicular bodies. Reduced MHC-II surface expression in S. Typhimurium-infected BMDCs correlated with reduced antigen presentation to Type A T cells. Salmonella infection is implicated in reactive arthritis. Therefore, polarisation of antigen presentation towards a Type B response by Salmonella may be a predisposing factor in autoimmune conditions such as reactive arthritis.
© 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures


Similar articles
-
Recognition of Salmonella by Dectin-1 induces presentation of peptide antigen to type B T cells.Eur J Immunol. 2014 Apr;44(4):962-9. doi: 10.1002/eji.201344065. Epub 2014 Feb 16. Eur J Immunol. 2014. PMID: 24458430 Free PMC article.
-
Salmonella infection of bone marrow-derived macrophages and dendritic cells: influence on antigen presentation and initiating an immune response.FEMS Immunol Med Microbiol. 2000 Apr;27(4):313-20. doi: 10.1111/j.1574-695X.2000.tb01445.x. FEMS Immunol Med Microbiol. 2000. PMID: 10727887 Review.
-
Identification of MHC-Bound Peptides from Dendritic Cells Infected with Salmonella enterica Strain SL1344: Implications for a Nontyphoidal Salmonella Vaccine.J Proteome Res. 2017 Jan 6;16(1):298-306. doi: 10.1021/acs.jproteome.6b00926. Epub 2016 Nov 10. J Proteome Res. 2017. PMID: 27802388 Free PMC article.
-
Bacterial antigen delivery systems: phagocytic processing of bacterial antigens for MHC-I and MHC-II presentation to T cells.Behring Inst Mitt. 1997 Feb;(98):197-211. Behring Inst Mitt. 1997. PMID: 9382741 Review.
-
Processing of viable Salmonella typhimurium for presentation of a CD4 T cell epitope from the Salmonella invasion protein C (SipC).Eur J Immunol. 2002 Sep;32(9):2664-71. doi: 10.1002/1521-4141(200209)32:9<2664::AID-IMMU2664>3.0.CO;2-N. Eur J Immunol. 2002. PMID: 12207351
Cited by
-
The Salmonella Effector SteD Mediates MARCH8-Dependent Ubiquitination of MHC II Molecules and Inhibits T Cell Activation.Cell Host Microbe. 2016 Nov 9;20(5):584-595. doi: 10.1016/j.chom.2016.10.007. Cell Host Microbe. 2016. PMID: 27832589 Free PMC article.
-
Antigen recognition in autoimmune diabetes: a novel pathway underlying disease initiation.Precis Clin Med. 2018 Dec;1(3):102-110. doi: 10.1093/pcmedi/pby015. Epub 2018 Nov 23. Precis Clin Med. 2018. PMID: 30687564 Free PMC article. Review.
-
Salmonella Persist in Activated Macrophages in T Cell-Sparse Granulomas but Are Contained by Surrounding CXCR3 Ligand-Positioned Th1 Cells.Immunity. 2018 Dec 18;49(6):1090-1102.e7. doi: 10.1016/j.immuni.2018.10.009. Epub 2018 Dec 11. Immunity. 2018. PMID: 30552021 Free PMC article.
-
Unconventional T-cell recognition of an arthritogenic epitope of proteoglycan aggrecan released from degrading cartilage.Immunology. 2016 Apr;147(4):389-98. doi: 10.1111/imm.12557. Epub 2015 Dec 21. Immunology. 2016. PMID: 26581676 Free PMC article.
-
Nature and consequences of interactions between Salmonella enterica serovar Dublin and host cells in cattle.Vet Res. 2019 Nov 27;50(1):99. doi: 10.1186/s13567-019-0720-5. Vet Res. 2019. PMID: 31771636 Free PMC article.
References
-
- Brumell JH, Grinstein S. Salmonella redirects phagosomal maturation. Curr. Opin. Microbiol. 2004;7:78–84. - PubMed
-
- Rescigno M, Urbano M, Valzasina B, Francolini M, Rotta G, Bonasio R, Granucci F, et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2001;2:361–367. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

