Nanoscale mechanism of molecular transport through the nuclear pore complex as studied by scanning electrochemical microscopy
- PMID: 23320434
- PMCID: PMC3572272
- DOI: 10.1021/ja311080j
Nanoscale mechanism of molecular transport through the nuclear pore complex as studied by scanning electrochemical microscopy
Abstract
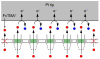
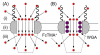
The nuclear pore complex (NPC) is the proteinaceous nanopore that solely mediates molecular transport across the nuclear envelope between the nucleus and cytoplasm of a eukaryotic cell. Small molecules (<40 kDa) diffuse through the large pore of this multiprotein complex. A passively impermeable macromolecule tagged with a signal peptide is chaperoned through the nanopore by nuclear transport receptors (e.g., importins) owing to their interactions with barrier-forming proteins. Presently, this bimodal transport mechanism is not well understood and is described by controversial models. Herein, we report on a dynamic and spatially resolved mechanism for NPC-mediated molecular transport through nanoscale central and peripheral routes with distinct permeabilities. Specifically, we develop a nanogap-based approach of scanning electrochemical microscopy to precisely measure the extremely high permeability of the nuclear envelope to a small probe molecule, (ferrocenylmethyl)trimethylammonium. Effective medium theories indicate that the passive permeability of 5.9 × 10(-2) cm/s corresponds to the free diffusion of the probe molecule through ~22 nanopores with a radius of 24 nm and a length of 35 nm. Peripheral routes are blocked by wheat germ agglutinin to yield 2-fold lower permeability for 17 nm-radius central routes. This lectin is also used in fluorescence assays to find that importins facilitate the transport of signal-tagged albumin mainly through the 7 nm-thick peripheral route rather than through the sufficiently large central route. We propose that this spatial selectivity is regulated by the conformational changes in barrier-forming proteins that transiently and locally expand the impermeably thin peripheral route while blocking the central route.
Figures






Similar articles
-
Nanoscale electrostatic gating of molecular transport through nuclear pore complexes as probed by scanning electrochemical microscopy.Chem Sci. 2019 Jul 8;10(34):7929-7936. doi: 10.1039/c9sc02356a. eCollection 2019 Sep 14. Chem Sci. 2019. PMID: 31673318 Free PMC article.
-
Nanoscale Quantitative Imaging of Single Nuclear Pore Complexes by Scanning Electrochemical Microscopy.Anal Chem. 2024 Jul 2;96(26):10765-10771. doi: 10.1021/acs.analchem.4c01890. Epub 2024 Jun 21. Anal Chem. 2024. PMID: 38904303 Free PMC article.
-
Nanoelectrochemical Study of Molecular Transport through the Nuclear Pore Complex.Chem Rec. 2021 Jun;21(6):1430-1441. doi: 10.1002/tcr.202000175. Epub 2021 Jan 27. Chem Rec. 2021. PMID: 33502100 Free PMC article. Review.
-
Characterisation of the passive permeability barrier of nuclear pore complexes.EMBO J. 2009 Sep 2;28(17):2541-53. doi: 10.1038/emboj.2009.200. Epub 2009 Aug 13. EMBO J. 2009. PMID: 19680228 Free PMC article.
-
Distinct, but not completely separate spatial transport routes in the nuclear pore complex.Nucleus. 2013 May-Jun;4(3):166-75. doi: 10.4161/nucl.24874. Epub 2013 May 1. Nucleus. 2013. PMID: 23669120 Free PMC article. Review.
Cited by
-
Single molecule fluorescence approaches shed light on intracellular RNAs.Chem Rev. 2014 Mar 26;114(6):3224-65. doi: 10.1021/cr400496q. Epub 2014 Jan 8. Chem Rev. 2014. PMID: 24417544 Free PMC article. Review. No abstract available.
-
Probing the disordered domain of the nuclear pore complex through coarse-grained molecular dynamics simulations.Biophys J. 2014 Sep 16;107(6):1393-402. doi: 10.1016/j.bpj.2014.07.060. Biophys J. 2014. PMID: 25229147 Free PMC article.
-
Nanoscale Hydrophobicity of Transport Barriers in the Nuclear Pore Complex as Compared with the Liquid/Liquid Interface by Scanning Electrochemical Microscopy.Anal Chem. 2025 Feb 11;97(5):2745-2753. doi: 10.1021/acs.analchem.4c04861. Epub 2025 Jan 29. Anal Chem. 2025. PMID: 39878353 Free PMC article.
-
Simple biophysics underpins collective conformations of the intrinsically disordered proteins of the Nuclear Pore Complex.Elife. 2016 May 20;5:e10785. doi: 10.7554/eLife.10785. Elife. 2016. PMID: 27198189 Free PMC article.
-
Nanoscale electrostatic gating of molecular transport through nuclear pore complexes as probed by scanning electrochemical microscopy.Chem Sci. 2019 Jul 8;10(34):7929-7936. doi: 10.1039/c9sc02356a. eCollection 2019 Sep 14. Chem Sci. 2019. PMID: 31673318 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

