Transgenic mice overexpressing neuregulin-1 model neurofibroma-malignant peripheral nerve sheath tumor progression and implicate specific chromosomal copy number variations in tumorigenesis
- PMID: 23321323
- PMCID: PMC3586689
- DOI: 10.1016/j.ajpath.2012.11.017
Transgenic mice overexpressing neuregulin-1 model neurofibroma-malignant peripheral nerve sheath tumor progression and implicate specific chromosomal copy number variations in tumorigenesis
Abstract
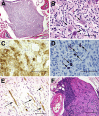
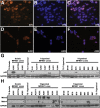
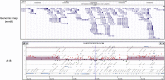
Patients with neurofibromatosis type 1 (NF1) develop benign plexiform neurofibromas that frequently progress to become malignant peripheral nerve sheath tumors (MPNSTs). A genetically engineered mouse model that accurately models plexiform neurofibroma-MPNST progression in humans would facilitate identification of somatic mutations driving this process. We previously reported that transgenic mice overexpressing the growth factor neuregulin-1 in Schwann cells (P(0)-GGFβ3 mice) develop MPNSTs. To determine whether P(0)-GGFβ3 mice accurately model human neurofibroma-MPNST progression, cohorts of these animals were monitored through death and were necropsied; 94% developed multiple neurofibromas, with 70% carrying smaller numbers of MPNSTs. Nascent MPNSTs were identified within neurofibromas, suggesting that these sarcomas arise from neurofibromas. Although neurofibromin expression was maintained, P(0)-GGFβ3 MPNSTs exhibited Ras hyperactivation, as in human NF1-associated MPNSTs. P(0)-GGFβ3 MPNSTs also exhibited abnormalities in the p16(INK4A)-cyclin D/CDK4-Rb and p19(ARF)-Mdm-p53 pathways, analogous to their human counterparts. Array comparative genomic hybridization (CGH) demonstrated reproducible chromosomal alterations in P(0)-GGFβ3 MPNST cells (including universal chromosome 11 gains) and focal gains and losses affecting 39 neoplasia-associated genes (including Pten, Tpd52, Myc, Gli1, Xiap, and Bbc3/PUMA). Array comparative genomic hybridization also identified recurrent focal copy number variations affecting genes not previously linked to neurofibroma or MPNST pathogenesis. We conclude that P(0)-GGFβ3 mice represent a robust model of neurofibroma-MPNST progression useful for identifying novel genes driving neurofibroma and MPNST pathogenesis.
Copyright © 2013 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures







References
-
- Gutmann D.H., Aylsworth A., Carey J.C., Korf B., Marks J., Pyeritz R.E., Rubenstein A., Viskochil D. The diagnostic evaluation and multidisciplinary management of neurofibromatosis 1 and neurofibromatosis 2. JAMA. 1997;278:51–57. - PubMed
-
- Carroll S.L., Stonecypher M.S. Tumor suppressor mutations and growth factor signaling in the pathogenesis of NF1-associated peripheral nerve sheath tumors. I. The role of tumor suppressor mutations. J Neuropathol Exp Neurol. 2004;63:1115–1123. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

