A new class of NO-donor pro-drugs triggered by γ-glutamyl transpeptidase with potential for reno-selective vasodilatation
- PMID: 23322269
- PMCID: PMC3785133
- DOI: 10.1039/c2cc38382a
A new class of NO-donor pro-drugs triggered by γ-glutamyl transpeptidase with potential for reno-selective vasodilatation
Abstract
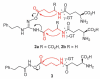
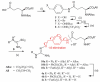
This communication describes the synthesis of a new class of N-hydroxyguanidine (NHG) pro-drugs which release nitric oxide (NO), triggered by the action of γ-glutamyl transpeptidase (γ-GT), and have potential for the treatment of acute renal injury/failure (ARI/ARF).
Figures
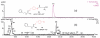
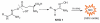


Similar articles
-
Development and characterization of glutamyl-protected N-hydroxyguanidines as reno-active nitric oxide donor drugs with therapeutic potential in acute renal failure.J Med Chem. 2013 Jul 11;56(13):5321-34. doi: 10.1021/jm400146r. Epub 2013 Jun 20. J Med Chem. 2013. PMID: 23782349
-
Stimulation of the hydrolytic activity and decrease of the transpeptidase activity of gamma-glutamyl transpeptidase by maleate; identity of a rat kidney maleate-stimulated glutaminase and gamma-glutamyl transpeptidase.Proc Natl Acad Sci U S A. 1974 Sep;71(9):3329-33. doi: 10.1073/pnas.71.9.3329. Proc Natl Acad Sci U S A. 1974. PMID: 4154442 Free PMC article.
-
Renal selective N-acetyl-gamma-glutamyl prodrugs. II. Carrier-mediated transport and intracellular conversion as determinants in the renal selectivity of N-acetyl-gamma-glutamyl sulfamethoxazole.J Pharmacol Exp Ther. 1990 Mar;252(3):1255-60. J Pharmacol Exp Ther. 1990. PMID: 1969473
-
[Nitrates and nitric oxide].Ital Heart J Suppl. 2000 Sep;1(9):1160-3. Ital Heart J Suppl. 2000. PMID: 11140284 Review. Italian.
-
New aspects of glutathione metabolism and translocation in mammals.Ciba Found Symp. 1979;(72):135-61. doi: 10.1002/9780470720554.ch9. Ciba Found Symp. 1979. PMID: 45011 Review.
Cited by
-
Glutathione S-conjugates as prodrugs to target drug-resistant tumors.Front Pharmacol. 2014 Aug 11;5:181. doi: 10.3389/fphar.2014.00181. eCollection 2014. Front Pharmacol. 2014. PMID: 25157234 Free PMC article. Review.
-
Nanoparticulation of Prodrug into Medicines for Cancer Therapy.Adv Sci (Weinh). 2021 Sep;8(18):e2101454. doi: 10.1002/advs.202101454. Epub 2021 Jul 29. Adv Sci (Weinh). 2021. PMID: 34323373 Free PMC article. Review.
References
-
- Bonvebtre JV, Weinberg JM. J. Am. Soc. Nephrol. 2003;14:2199–2210. - PubMed
-
- Sutton TA, Fisher CJ, Molitoris BA. Kidney Int. 2002;62:1539–1549. - PubMed
-
- Mariscalco G, Lorusso R, Dominici C, Renzulli A, Sala A. Ann. Thorac. Surg. 2011;92:1539–1547. - PubMed
-
- Grace PA. Br. J. Surg. 1994;81:637–647. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

