Temporal transcriptional response during infection of type II alveolar epithelial cells with Francisella tularensis live vaccine strain (LVS) supports a general host suppression and bacterial uptake by macropinocytosis
- PMID: 23322778
- PMCID: PMC3624459
- DOI: 10.1074/jbc.M112.362178
Temporal transcriptional response during infection of type II alveolar epithelial cells with Francisella tularensis live vaccine strain (LVS) supports a general host suppression and bacterial uptake by macropinocytosis
Abstract
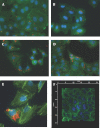
Pneumonic tularemia is caused by inhalation of Francisella tularensis, one of the most infectious microbes known. We wanted to study the kinetics of the initial and early interactions between bacterium and host cells in the lung. To do this, we examined the infection of A549 airway epithelial cells with the live vaccine strain (LVS) of F. tularensis. A549 cells were infected and analyzed for global transcriptional response at multiple time points up to 16 h following infection. At 15 min and 2 h, a strong transcriptional response was observed including cytoskeletal rearrangement, intracellular transport, and interferon signaling. However, at later time points (6 and 16 h), very little differential gene expression was observed, indicating a general suppression of the host response consistent with other reported cell lines and murine tissues. Genes for macropinocytosis and actin/cytoskeleton rearrangement were highly up-regulated and common to the 15 min and 2 h time points, suggesting the use of this method for bacterial entry into cells. We demonstrate macropinocytosis through the uptake of FITC-dextran and amiloride inhibition of Francisella LVS uptake. Our results suggest that macropinocytosis is a potential mechanism of intracellular entry by LVS and that the host cell response is suppressed during the first 2-6 h of infection. These results suggest that the attenuated Francisella LVS induces significant host cell signaling at very early time points after the bacteria's interaction with the cell.
Figures
Similar articles
-
Role of primary human alveolar epithelial cells in host defense against Francisella tularensis infection.Infect Immun. 2007 Aug;75(8):3969-78. doi: 10.1128/IAI.00157-07. Epub 2007 May 14. Infect Immun. 2007. PMID: 17502386 Free PMC article.
-
Immunologic consequences of Francisella tularensis live vaccine strain infection: role of the innate immune response in infection and immunity.J Immunol. 2006 Jun 1;176(11):6888-99. doi: 10.4049/jimmunol.176.11.6888. J Immunol. 2006. PMID: 16709849
-
Francisella tularensis live vaccine strain folate metabolism and pseudouridine synthase gene mutants modulate macrophage caspase-1 activation.Infect Immun. 2013 Jan;81(1):201-8. doi: 10.1128/IAI.00991-12. Epub 2012 Oct 31. Infect Immun. 2013. PMID: 23115038 Free PMC article.
-
Innate and adaptive immune responses to an intracellular bacterium, Francisella tularensis live vaccine strain.Microbes Infect. 2003 Feb;5(2):135-42. doi: 10.1016/s1286-4579(02)00084-9. Microbes Infect. 2003. PMID: 12650771 Review.
-
Nature of protective immunity to Francisella tularensis.Rev Infect Dis. 1989 May-Jun;11(3):440-51. Rev Infect Dis. 1989. PMID: 2665002 Review.
Cited by
-
Water-pipe smoke condensate increases the internalization of Mycobacterium Bovis of type II alveolar epithelial cells (A549).BMC Pulm Med. 2017 Apr 21;17(1):68. doi: 10.1186/s12890-017-0413-7. BMC Pulm Med. 2017. PMID: 28431548 Free PMC article.
-
Interactions of Francisella tularensis with Alveolar Type II Epithelial Cells and the Murine Respiratory Epithelium.PLoS One. 2015 May 26;10(5):e0127458. doi: 10.1371/journal.pone.0127458. eCollection 2015. PLoS One. 2015. PMID: 26010977 Free PMC article.
-
A Francisella novicida Mutant, Lacking the Soluble Lytic Transglycosylase Slt, Exhibits Defects in Both Growth and Virulence.Front Microbiol. 2019 Jun 14;10:1343. doi: 10.3389/fmicb.2019.01343. eCollection 2019. Front Microbiol. 2019. PMID: 31258523 Free PMC article.
-
Francisella philomiragia Infection and Lethality in Mammalian Tissue Culture Cell Models, Galleria mellonella, and BALB/c Mice.Front Microbiol. 2016 May 24;7:696. doi: 10.3389/fmicb.2016.00696. eCollection 2016. Front Microbiol. 2016. PMID: 27252681 Free PMC article.
-
A novel method of Francisella infection of epithelial cells using HeLa cells expressing fc gamma receptor.BMC Infect Dis. 2024 Oct 17;24(1):1171. doi: 10.1186/s12879-024-10083-y. BMC Infect Dis. 2024. PMID: 39420255 Free PMC article.
References
-
- Dennis D. T., Inglesby T. V., Henderson D. A., Bartlett J. G., Ascher M. S., Eitzen E., Fine A. D., Friedlander A. M., Hauer J., Layton M., Lillibridge S. R., McDade J. E., Osterholm M. T., O'Toole T., Parker G., Perl T. M., Russell P. K., Tonat K., and Working Group on Civilian Biodefense (2001) Tularemia as a Biological Weapon. Medical and Public Health Management. JAMA 285, 2763–2773 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

