BAFF/APRIL system in pediatric OMS: relation to severity, neuroinflammation, and immunotherapy
- PMID: 23324534
- PMCID: PMC3610127
- DOI: 10.1186/1742-2094-10-10
BAFF/APRIL system in pediatric OMS: relation to severity, neuroinflammation, and immunotherapy
Abstract
Background: B-cell dysregulation has been implicated but not fully characterized in pediatric opsoclonus-myoclonus syndrome (OMS), a neuroblastoma-associated neuroinflammatory disorder.
Objective: To assess the role of B-cell activating factor (BAFF) and a proliferation-inducing ligand (APRIL), two critical B cell-modulating cytokines, as potential biomarkers of disease activity and treatment biomarkers in OMS.
Methods: Soluble BAFF and APRIL were measured in cerebrospinal fluid (CSF) and serum by ELISA in 433 children (296 OMS, 109 controls, 28 other inflammatory neurological disorders (OIND)). BAFF-R receptors on circulating CD19+ B cells were measured by flow cytometry. A blinded scorer rated motor severity on the OMS Evaluation Scale. Immunotherapies were evaluated cross-sectionally and longitudinally.
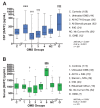
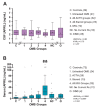
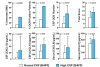
Results: The mean CSF BAFF concentration, which was elevated in untreated OMS and OIND, correlated with OMS severity category (P = 0.006), and reduction by adrenocorticotropic hormone or corticotropin (ACTH) (-61%) or corticosteroids (-38%) was seen at each level of severity. In contrast, CSF APRIL was normal in OMS and OIND and unaffected by immunotherapy. When the entire OMS dataset was dichotomized into 'high' versus 'normal' CSF BAFF concentration, the phenotype of the high group included greater motor severity and number of CSF oligoclonal bands, and a higher concentration of inflammatory chemokines CXCL13 and CXCL10 in CSF and CXCL9 and CCL21 in serum. Serum APRIL was 6.7-fold higher in the intravenous immunoglobulins (IVIg) group, whereas serum BAFF was 2.6-fold higher in the rituximab group. The frequency of B cell BAFF-R expression was similar in untreated and treated OMS. Longitudinal studies of CSF BAFF revealed a significant decline in ACTH-treated patients (with or without rituximab) (P < 0.0001). Longitudinal studies of serum APRIL showed a 2.9-fold increase after 1 to 2 g/kg IVIg monotherapy (P = 0.0003).
Conclusions: Striking distinctions in BAFF/APRIL signaling were found. OMS displayed heterogeneity in CSF BAFF expression, which met many but not all criteria as a potential biomarker of disease activity. We speculate that CSF BAFF may have more utility in a biomarker panel than as a stand-alone biomarker, and that the selective upregulation of both serum APRIL by IVIg and BAFF by rituximab, as well as downregulation of CSF BAFF by ACTH/steroids, may have utility as treatment biomarkers.
Figures
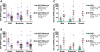
References
-
- Tate ED, Pranzatelli MR, Verhulst SJ, Markwell SJ, Franz DN, Graf WD, Joseph SA, Khakoo YN, Lo WD, Mitchell WG, Sivaswamy L. Active comparator-controlled, rater-blinded study of corticotropin-based immunotherapies for opsoclonus-myoclonus syndrome. J Child Neurol. 2012;27:875–884. doi: 10.1177/0883073811428816. - DOI - PubMed
-
- Pranzatelli MR, Tate ED, Hoefgen ER, Verhulst SJ. Interferon-inducible protein 10 (CXCL10) is increased in the cerebrospinal fluid of children with opsoclonus-myoclonus syndrome. Ann Neurol. 2007;Suppl 11:S112–S113.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

