Wnt5a cooperates with canonical Wnts to generate midbrain dopaminergic neurons in vivo and in stem cells
- PMID: 23324743
- PMCID: PMC3574960
- DOI: 10.1073/pnas.1208524110
Wnt5a cooperates with canonical Wnts to generate midbrain dopaminergic neurons in vivo and in stem cells
Abstract
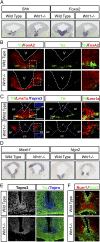
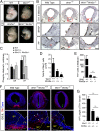
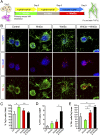
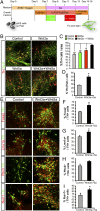
Wnts are a family of secreted proteins that regulate multiple steps of neural development and stem cell differentiation. Two of them, Wnt1 and Wnt5a, activate distinct branches of Wnt signaling and individually regulate different aspects of midbrain dopaminergic (DA) neuron development. However, several of their functions and interactions remain to be elucidated. Here, we report that loss of Wnt1 results in loss of Lmx1a and Ngn2 expression, as well as agenesis of DA neurons in the midbrain floor plate. Remarkably, a few ectopic DA neurons still emerge in the basal plate of Wnt1(-/-) mice, where Lmx1a is ectopically expressed. These results indicate that Wnt1 orchestrates DA specification and neurogenesis in vivo. Analysis of Wnt1(-/-);Wnt5a(-/-) mice revealed a greater loss of Nurr1(+) cells and DA neurons than in single mutants, indicating that Wnt1 and Wnt5a interact genetically and cooperate to promote midbrain DA neuron development in vivo. Our results unravel a functional interaction between Wnt1 and Wnt5a resulting in enhanced DA neurogenesis. Taking advantage of these findings, we have developed an application of Wnts to improve the generation of midbrain DA neurons from neural and embryonic stem cells. We thus show that coordinated Wnt actions promote DA neuron development in vivo and in stem cells and suggest that coordinated Wnt administration can be used to improve DA differentiation of stem cells and the development of stem cell-based therapies for Parkinson's disease.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Sugimura R, Li L. Noncanonical Wnt signaling in vertebrate development, stem cells, and diseases. Birth Defects Res C Embryo Today. 2010;90(4):243–256. - PubMed
-
- Inestrosa NC, Arenas E. Emerging roles of Wnts in the adult nervous system. Nat Rev Neurosci. 2010;11(2):77–86. - PubMed
-
- Angers S, Moon RT. Proximal events in Wnt signal transduction. Nat Rev Mol Cell Biol. 2009;10(7):468–477. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

