Icosapent ethyl, a pure ethyl ester of eicosapentaenoic acid: effects on circulating markers of inflammation from the MARINE and ANCHOR studies
- PMID: 23325450
- PMCID: PMC3572383
- DOI: 10.1007/s40256-012-0002-3
Icosapent ethyl, a pure ethyl ester of eicosapentaenoic acid: effects on circulating markers of inflammation from the MARINE and ANCHOR studies
Abstract
Background: Icosapent ethyl (IPE) is a high-purity prescription form of eicosapentaenoic acid ethyl ester approved by the US Food and Drug Administration as an adjunct to diet to reduce triglyceride (TG) levels in adult patients with severe (≥500 mg/dL) hypertriglyceridemia. In addition to TG-lowering effects, IPE also reduces non-high-density lipoprotein cholesterol and apolipoprotein B levels without significantly increasing low-density lipoprotein cholesterol (LDL-C) in patients with very high TG levels ≥500 mg/dL (MARINE study) and in patients with well-controlled LDL-C and residually high TG levels 200-500 mg/dL (ANCHOR study). This analysis examined the effect of IPE on inflammatory markers in patients from MARINE and ANCHOR.
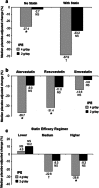
Methods: MARINE (N = 229) and ANCHOR (N = 702) were Phase III, double-blind studies that randomized hypertriglyceridemic patients to IPE 4 g/day, 2 g/day, or placebo. This analysis assessed the median placebo-adjusted percentage change from baseline in markers representing various stages of atherosclerotic inflammation such as intercellular adhesion molecule-1 (ICAM-1), oxidized low-density lipoprotein (Ox-LDL), lipoprotein-associated phospholipase A(2) (Lp-PLA(2)), interleukin-6 (IL-6), and high-sensitivity C-reactive protein (hsCRP).
Results: Compared to placebo, IPE 4 g/day significantly decreased Ox-LDL (13 %, p < 0.0001, ANCHOR), Lp-PLA(2) (14 %, p < 0.001, MARINE; 19 %, p < 0.0001, ANCHOR), and hsCRP levels (36 %, p < 0.01, MARINE; 22 %, p < 0.001, ANCHOR), but did not significantly change ICAM-1 and IL-6 levels. In the MARINE study, IPE 2 g/day did not significantly change ICAM-1, Ox-LDL, Lp-PLA(2), IL-6, or hsCRP levels. Also, compared to placebo in the ANCHOR study, IPE 2 g/day significantly decreased Lp-PLA(2) levels (8 %, p < 0.0001), but did not significantly change levels of other assessed inflammatory markers.
Conclusion: Compared to placebo, in hypertriglyceridemic patients, IPE 4 g/day significantly decreased Ox-LDL, Lp-PLA(2), and hsCRP levels.
Figures

References
-
- Hwang SJ, Ballantyne CM, Sharrett AR, et al. Circulating adhesion molecules VCAM-1, ICAM-1, and E-selectin in carotid atherosclerosis and incident coronary heart disease cases: the Atherosclerosis Risk In Communities (ARIC) study. Circulation. 1997;96(12):4219–4225. doi: 10.1161/01.CIR.96.12.4219. - DOI - PubMed
-
- Lawson C, Wolf S. ICAM-1 signaling in endothelial cells. Pharmacol Rep. 2009;61(1):22–32. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

