Collagen remodeling by phagocytosis is determined by collagen substrate topology and calcium-dependent interactions of gelsolin with nonmuscle myosin IIA in cell adhesions
- PMID: 23325791
- PMCID: PMC3596245
- DOI: 10.1091/mbc.E12-10-0754
Collagen remodeling by phagocytosis is determined by collagen substrate topology and calcium-dependent interactions of gelsolin with nonmuscle myosin IIA in cell adhesions
Abstract
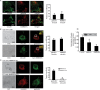
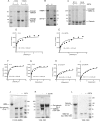
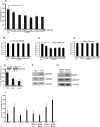
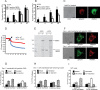
We examine how collagen substrate topography, free intracellular calcium ion concentration ([Ca(2+)]i, and the association of gelsolin with nonmuscle myosin IIA (NMMIIA) at collagen adhesions are regulated to enable collagen phagocytosis. Fibroblasts plated on planar, collagen-coated substrates show minimal increase of [Ca(2+)]i, minimal colocalization of gelsolin and NMMIIA in focal adhesions, and minimal intracellular collagen degradation. In fibroblasts plated on collagen-coated latex beads there are large increases of [Ca(2+)]i, time- and Ca(2+)-dependent enrichment of NMMIIA and gelsolin at collagen adhesions, and abundant intracellular collagen degradation. NMMIIA knockdown retards gelsolin recruitment to adhesions and blocks collagen phagocytosis. Gelsolin exhibits tight, Ca(2+)-dependent binding to full-length NMMIIA. Gelsolin domains G4-G6 selectively require Ca(2+) to interact with NMMIIA, which is restricted to residues 1339-1899 of NMMIIA. We conclude that cell adhesion to collagen presented on beads activates Ca(2+) entry and promotes the formation of phagosomes enriched with NMMIIA and gelsolin. The Ca(2+) -dependent interaction of gelsolin and NMMIIA in turn enables actin remodeling and enhances collagen degradation by phagocytosis.
Figures


References
-
- Arora PD, Bibby KJ, McCulloch CA. Slow oscillations of free intracellular calcium ion concentration in human fibroblasts responding to mechanical stretch. J Cell Physiol. 1994;161:187–200. - PubMed
-
- Arora PD, Fan L, Sodek J, Kapus A, McCulloch CA. Differential binding to dorsal and ventral cell surfaces of fibroblasts: effect on collagen phagocytosis. Exp Cell Res. 2003;286:366–380. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

