UNC93B1 and nucleic acid-sensing Toll-like receptors mediate host resistance to infection with Leishmania major
- PMID: 23325805
- PMCID: PMC3591622
- DOI: 10.1074/jbc.M112.407684
UNC93B1 and nucleic acid-sensing Toll-like receptors mediate host resistance to infection with Leishmania major
Abstract
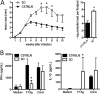
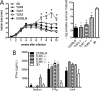
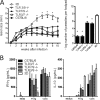
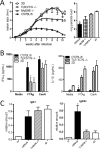
The mammalian homolog B1 of Unc-93 Caenorhabditis elegans known as UNC93B1 is a chaperone protein that mediates translocation of the nucleic acid-sensing Toll-like receptors (TLRs) from the endoplasmic reticulum to the endolysosomes. The triple deficient (UNC93B1 mutant) mice have a functional single point mutation in the UNC93B1 that results in non-functional TLR3, TLR7, and TLR9. Herein, we demonstrate that UNC93B1 mutant mice, in the C57BL/6 (resistant) genetic background, are highly susceptible to Leishmania major infection. Enhanced swelling of the footpad was associated with high levels of interleukin 10, decreased levels of interferon γ, and increased parasitism. None of the single TLR3, TLR7, and TLR9 knock-out (KO) mice resemble the UNC93B1 mutant phenotype upon infection with L. major. Whereas the double TLR7/TLR9 KO showed a partial phenotype, the triple TLR3/TLR7/TLR9 KO mice were as susceptible as the UNC93B1 mutant mice, when infected with Leishmania parasites. Finally, we demonstrate that treatment with either anti-interleukin 10 receptor monoclonal antibody or recombinant interleukin 12 restored a robust anti-parasite TH1 response and reverted the susceptible phenotype of UNC93B1 mutant mice. Altogether, our results indicate the redundant and essential role of nucleic acid-sensing TLR3, TLR7 and TLR9 in inducing interleukin 12, development of a TH1 response, and resistance to L. major infection in resistant C57BL/6 mice.
Figures




Similar articles
-
Autophagy downstream of endosomal Toll-like receptor signaling in macrophages is a key mechanism for resistance to Leishmania major infection.J Biol Chem. 2017 Aug 11;292(32):13087-13096. doi: 10.1074/jbc.M117.780981. Epub 2017 Jun 12. J Biol Chem. 2017. PMID: 28607148 Free PMC article.
-
Unc93B1 restricts systemic lethal inflammation by orchestrating Toll-like receptor 7 and 9 trafficking.Immunity. 2011 Jul 22;35(1):69-81. doi: 10.1016/j.immuni.2011.05.010. Epub 2011 Jun 16. Immunity. 2011. PMID: 21683627
-
Unc93B1 biases Toll-like receptor responses to nucleic acid in dendritic cells toward DNA- but against RNA-sensing.J Exp Med. 2009 Jun 8;206(6):1339-50. doi: 10.1084/jem.20082316. Epub 2009 May 18. J Exp Med. 2009. PMID: 19451267 Free PMC article.
-
Regulatory molecules required for nucleotide-sensing Toll-like receptors.Immunol Rev. 2009 Jan;227(1):32-43. doi: 10.1111/j.1600-065X.2008.00729.x. Immunol Rev. 2009. PMID: 19120473 Review.
-
Cell Surface Expression of Endosomal Toll-Like Receptors-A Necessity or a Superfluous Duplication?Front Immunol. 2021 Feb 1;11:620972. doi: 10.3389/fimmu.2020.620972. eCollection 2020. Front Immunol. 2021. PMID: 33597952 Free PMC article. Review.
Cited by
-
Do you see what I see: Recognition of protozoan parasites by Toll-like receptors.Curr Immunol Rev. 2013 Aug;9(3):129-140. doi: 10.2174/1573395509666131203225929. Curr Immunol Rev. 2013. PMID: 25383072 Free PMC article.
-
CNBP controls IL-12 gene transcription and Th1 immunity.J Exp Med. 2018 Dec 3;215(12):3136-3150. doi: 10.1084/jem.20181031. Epub 2018 Nov 15. J Exp Med. 2018. PMID: 30442645 Free PMC article.
-
Donor UNC-93 Homolog B1 genetic polymorphism predicts survival outcomes after unrelated bone marrow transplantation.Genes Immun. 2021 May;22(1):35-43. doi: 10.1038/s41435-021-00122-y. Epub 2021 Feb 24. Genes Immun. 2021. PMID: 33627833 Free PMC article.
-
Macrophage Mitochondrial Biogenesis and Metabolic Reprogramming Induced by Leishmania donovani Require Lipophosphoglycan and Type I Interferon Signaling.mBio. 2022 Dec 20;13(6):e0257822. doi: 10.1128/mbio.02578-22. Epub 2022 Oct 12. mBio. 2022. PMID: 36222510 Free PMC article.
-
Innate sensing of malaria parasites.Nat Rev Immunol. 2014 Nov;14(11):744-57. doi: 10.1038/nri3742. Epub 2014 Oct 17. Nat Rev Immunol. 2014. PMID: 25324127 Review.
References
-
- Sacks D., Noben-Trauth N. (2002) The immunology of susceptibility and resistance to Leishmania major in mice. Nat. Rev. Immunol. 2, 845–858 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

