Effectiveness of 5-flurouracil-based neoadjuvant chemotherapy in locally-advanced gastric/gastroesophageal cancer: a meta-analysis
- PMID: 23326149
- PMCID: PMC3544046
- DOI: 10.3748/wjg.v18.i48.7384
Effectiveness of 5-flurouracil-based neoadjuvant chemotherapy in locally-advanced gastric/gastroesophageal cancer: a meta-analysis
Abstract
Aim: To investigate the effectiveness of 5-flurouracil-based neoadjuvant chemotherapy (NAC) for gastroesophageal and gastric cancer by meta-analysis.
Methods: MEDLINE and manual searches were performed to identify all published randomized controlled trials (RCTs) investigating the efficacy of the flurouracil-based NAC for gastroesophageal and gastric cancer, and RCTs of NAC for advanced gastroesophageal and gastric cancer vs no therapy before surgery. Studies that included patients with metastases at enrollment were excluded. Primary endpoint was the odds ratio (OR) for improving overall survival rate of patients with gastroesophageal and gastric cancer. Secondary endpoints were the OR of efficiency for down-staging tumor and increasing R0 resection in patients with gastroesophageal and gastric cancer. Safety analyses were also performed. The OR was the principal measurement of effect, which was calculated as the treatment group (NAC plus surgery) vs control group (surgery alone) and was presented as a point estimate with 95% confidence intervals (CI). All calculations and statistical tests were performed using RevMan 5.1 software.
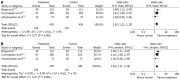
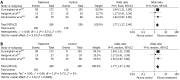
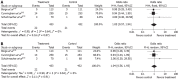
Results: Seven RCTs were included for the analysis. A total of 1249 patients with advanced gastroesophageal and gastric cancer enrolled in the seven trials were divided into treatment group (n = 620) and control group (n = 629). The quality scores of the RCTs were assessed according to the method of Jadad. The RCT quality scores ranged from 2 to 7 (5-point scale), with a mean of 3.75. The median follow-up time in these studies was over 3 years. The meta-analysis showed that NAC improved the overall survival rate (OR 1.40, 95%CI 1.11-1.76; P = 0.005), which was statistically significant. The 3-year progression-free survival rate was significantly higher in treatment group than in control group (37.7% vs 27.3%) (OR 1.62, 95%CI 1.21-2.15; P = 0.001). The tumor down-stage rate was higher in treatment group than in control group (55.76% vs 41.38%) (OR 1.77, 95%CI 1.27-2.49; P = 0.0009) and the R0 resection rate of the gastroesophageal and gastric cancer was higher in treatment group than in control group (75.11% vs 68.56%) (OR 1.38, 95%CI 1.03-1.85; P = 0.03), with significant differences. No obvious safety concerns about mortality and complications were raised in these trials. There were no statistically significant differences in perioperative mortality (5.08% vs 4.86%) (OR 1.05, 95%CI 0.57-1.94; P = 0.87 fixed-effect model) and in the complication rate between the two groups (13.25% vs 9.66%) (OR 1.40, 95%CI 0.91-2.14; P = 0.12 fixed-effect model). Trials showed that patients from Western countries favored NAC compared with those from Asian countries (OR 1.40, 95%CI 1.07-1.83). Monotherapy was inferior to multiple chemotherapy (OR 1.40, 95%CI 1.07-1.83). Intravenous administration of NAC was more advantageous than oral route (OR 1.41, 95%CI 1.09-1.81).
Conclusion: Flurouracil-based NAC can safely improve overall survival rate of patients with gastroesophageal/gastric cancer. Additionally, NAC can down the tumor stage and improve R0 resection.
Keywords: Gastric cancer; Gastroesophageal cancer; Meta-analysis; Neoadjuvant chemotherapy.
Figures





Similar articles
-
Neoadjuvant chemotherapy for advanced gastric cancer: a meta-analysis.World J Gastroenterol. 2010 Nov 28;16(44):5621-8. doi: 10.3748/wjg.v16.i44.5621. World J Gastroenterol. 2010. PMID: 21105197 Free PMC article.
-
Clinical effectiveness of neoadjuvant chemotherapy in advanced gastric cancer: an updated meta-analysis of randomized controlled trials.Eur J Surg Oncol. 2014 Oct;40(10):1321-30. doi: 10.1016/j.ejso.2014.01.006. Epub 2014 Feb 25. Eur J Surg Oncol. 2014. Retraction in: Eur J Surg Oncol. 2015 Jul;41(7):953. doi: 10.1016/j.ejso.2015.05.001. PMID: 25239442 Retracted.
-
An updated meta-analysis of randomized controlled trial assessing the effect of neoadjuvant chemotherapy in advanced gastric cancer.Cancer Invest. 2014 Jul;32(6):272-84. doi: 10.3109/07357907.2014.911877. Epub 2014 May 6. Cancer Invest. 2014. PMID: 24800782
-
A meta-analysis of randomized controlled trials that compared neoadjuvant chemotherapy and surgery to surgery alone for resectable esophageal cancer.Am J Surg. 2002 Mar;183(3):274-9. doi: 10.1016/s0002-9610(02)00795-x. Am J Surg. 2002. PMID: 11943125
-
Neoadjuvant chemotherapy for gastric cancer. Is it a must or a fake?World J Gastroenterol. 2018 Jan 14;24(2):274-289. doi: 10.3748/wjg.v24.i2.274. World J Gastroenterol. 2018. PMID: 29375213 Free PMC article. Review.
Cited by
-
Preoperative chemotherapy in gastric cancer: expanding the indications, limiting the overuse.Gastric Cancer. 2015 Jan;18(1):200-1. doi: 10.1007/s10120-014-0359-x. Epub 2014 Mar 29. Gastric Cancer. 2015. PMID: 24682591 No abstract available.
-
Case Report: Clinical application of continuous arterial infusion chemotherapy in neoadjuvant therapy for locally advanced gastric cancer.Front Oncol. 2023 Jun 23;13:1214599. doi: 10.3389/fonc.2023.1214599. eCollection 2023. Front Oncol. 2023. PMID: 37427136 Free PMC article.
-
Effect of neoadjuvant chemotherapy in patients with gastric cancer: a PRISMA-compliant systematic review and meta-analysis.BMC Cancer. 2018 Jan 31;18(1):118. doi: 10.1186/s12885-018-4027-0. BMC Cancer. 2018. PMID: 29385994 Free PMC article.
-
The optimal neoadjuvant chemotherapy regimen for locally advanced gastric and gastroesophageal junction adenocarcinoma: a systematic review and Bayesian network meta-analysis.Eur J Med Res. 2022 Nov 9;27(1):239. doi: 10.1186/s40001-022-00878-7. Eur J Med Res. 2022. PMID: 36352476 Free PMC article.
-
Pathological complete response and long-term survival in a very elderly patient after neoadjuvant chemotherapy for locally advanced, unresectable gastric cancer.Case Rep Oncol Med. 2014;2014:532924. doi: 10.1155/2014/532924. Epub 2014 Sep 14. Case Rep Oncol Med. 2014. PMID: 25298899 Free PMC article.
References
-
- Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150. - PubMed
-
- Botterweck AA, Schouten LJ, Volovics A, Dorant E, van Den Brandt PA. Trends in incidence of adenocarcinoma of the oesophagus and gastric cardia in ten European countries. Int J Epidemiol. 2000;29:645–654. - PubMed
-
- Nashimoto A, Nakajima T, Furukawa H, Kitamura M, Kinoshita T, Yamamura Y, Sasako M, Kunii Y, Motohashi H, Yamamoto S. Randomized trial of adjuvant chemotherapy with mitomycin, Fluorouracil, and Cytosine arabinoside followed by oral Fluorouracil in serosa-negative gastric cancer: Japan Clinical Oncology Group 9206-1. J Clin Oncol. 2003;21:2282–2287. - PubMed
-
- Briasoulis E, Liakakos T, Dova L, Fatouros M, Tsekeris P, Roukos DH, Kappas AM. Selecting a specific pre- or postoperative adjuvant therapy for individual patients with operable gastric cancer. Expert Rev Anticancer Ther. 2006;6:931–939. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

