Urinary proteomic biomarkers for diagnosis and risk stratification of autosomal dominant polycystic kidney disease: a multicentric study
- PMID: 23326375
- PMCID: PMC3542378
- DOI: 10.1371/journal.pone.0053016
Urinary proteomic biomarkers for diagnosis and risk stratification of autosomal dominant polycystic kidney disease: a multicentric study
Erratum in
- PLoS One. 2013;8(8). doi:10.1371/annotation/9281c713-d253-4a1a-8255-92e691e77a24
Abstract
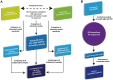
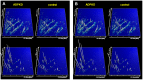
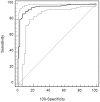
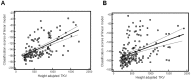
Treatment options for autosomal dominant polycystic kidney disease (ADPKD) will likely become available in the near future, hence reliable diagnostic and prognostic biomarkers for the disease are strongly needed. Here, we aimed to define urinary proteomic patterns in ADPKD patients, which aid diagnosis and risk stratification. By capillary electrophoresis online coupled to mass spectrometry (CE-MS), we compared the urinary peptidome of 41 ADPKD patients to 189 healthy controls and identified 657 peptides with significantly altered excretion, of which 209 could be sequenced using tandem mass spectrometry. A support-vector-machine based diagnostic biomarker model based on the 142 most consistent peptide markers achieved a diagnostic sensitivity of 84.5% and specificity of 94.2% in an independent validation cohort, consisting of 251 ADPKD patients from five different centers and 86 healthy controls. The proteomic alterations in ADPKD included, but were not limited to markers previously associated with acute kidney injury (AKI). The diagnostic biomarker model was highly specific for ADPKD when tested in a cohort consisting of 481 patients with a variety of renal and extrarenal diseases, including AKI. Similar to ultrasound, sensitivity and specificity of the diagnostic score depended on patient age and genotype. We were furthermore able to identify biomarkers for disease severity and progression. A proteomic severity score was developed to predict height adjusted total kidney volume (htTKV) based on proteomic analysis of 134 ADPKD patients and showed a correlation of r = 0.415 (p<0.0001) with htTKV in an independent validation cohort consisting of 158 ADPKD patients. In conclusion, the performance of peptidomic biomarker scores is superior to any other biochemical markers of ADPKD and the proteomic biomarker patterns are a promising tool for prognostic evaluation of ADPKD.
Conflict of interest statement
Figures
References
-
- Dalgaard OZ (1957) Bilateral polycystic disease of the kidneys; a follow-up of two hundred and eighty-four patients and their families. Acta Med Scand (Suppl 328) 1–255. - PubMed
-
- Iglesias CG, Torres VE, Offord KP, Holley KE, Beard CM, et al. (1983) Epidemiology of adult polycystic kidney disease, Olmsted County, Minnesota: 1935–1980. Am J Kidney Dis 2: 630–639. - PubMed
-
- Hateboer N, v Dijk MA, Bogdanova N, Coto E, Saggar-Malik AK, et al. (1999) Comparison of phenotypes of polycystic kidney disease types 1 and 2. European PKD1-PKD2 Study Group. Lancet 353: 103–107. - PubMed
-
- Chapman AB, Johnson AM, Gabow PA, Schrier RW (1994) Overt proteinuria and microalbuminuria in autosomal dominant polycystic kidney disease. J Am Soc Nephrol 5: 1349–1354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

