Desmoglein 2 is less important than desmoglein 3 for keratinocyte cohesion
- PMID: 23326495
- PMCID: PMC3543261
- DOI: 10.1371/journal.pone.0053739
Desmoglein 2 is less important than desmoglein 3 for keratinocyte cohesion
Abstract
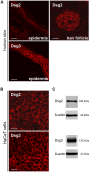
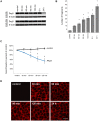
Desmosomes provide intercellular adhesive strength required for integrity of epithelial and some non-epithelial tissues. Within the epidermis, the cadherin-type adhesion molecules desmoglein (Dsg) 1-4 and desmocollin (Dsc) 1-3 build the adhesive core of desmosomes. In keratinocytes, several isoforms of these proteins are co-expressed. However, the contribution of specific isoforms to overall cell cohesion is unclear. Therefore, in this study we investigated the roles of Dsg2 and Dsg3, the latter of which is known to be essential for keratinocyte adhesion based on its autoantibody-induced loss of function in the autoimmune blistering skin disease pemphigus vulgaris (PV). The pathogenic PV antibody AK23, targeting the Dsg3 adhesive domain, led to profound loss of cell cohesion in human keratinocytes as revealed by the dispase-based dissociation assays. In contrast, an antibody against Dsg2 had no effect on cell cohesion although the Dsg2 antibody was demonstrated to interfere with Dsg2 transinteraction by single molecule atomic force microscopy and was effective to reduce cell cohesion in intestinal epithelial Caco-2 cells which express Dsg2 as the only Dsg isoform. To substantiate these findings, siRNA-mediated silencing of Dsg2 or Dsg3 was performed in keratinocytes. In contrast to Dsg3-depleted cells, Dsg2 knockdown reduced cell cohesion only under conditions of increased shear. These experiments indicate that specific desmosomal cadherins contribute differently to keratinocyte cohesion and that Dsg2 compared to Dsg3 is less important in this context.
Conflict of interest statement
Figures



References
-
- Getsios S, Huen AC, Green KJ (2004) Working out the strength and flexibility of desmosomes. Nat Rev Mol Cell Biol 5: 271–281. - PubMed
-
- Tsang SM, Brown L, Lin K, Liu L, Piper K, et al. (2012) Non-junctional human desmoglein 3 acts as an upstream regulator of Src in E-cadherin adhesion, a pathway possibly involved in the pathogenesis of pemphigus vulgaris. J Pathol 227: 81–93. - PubMed
-
- Stanley JR, Amagai M (2006) Pemphigus, bullous impetigo, and the staphylococcal scalded-skin syndrome. N Engl J Med 355: 1800–1810. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

