Hepatic stellate cells and portal fibroblasts are the major cellular sources of collagens and lysyl oxidases in normal liver and early after injury
- PMID: 23328207
- PMCID: PMC3602686
- DOI: 10.1152/ajpgi.00222.2012
Hepatic stellate cells and portal fibroblasts are the major cellular sources of collagens and lysyl oxidases in normal liver and early after injury
Abstract
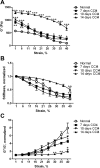
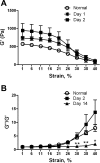
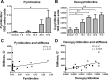
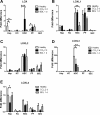
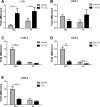
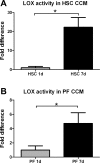
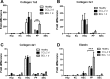
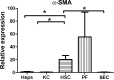
Liver fibrosis is characterized by excessive deposition of extracellular matrix proteins by myofibroblasts derived from hepatic stellate cells and portal fibroblasts. Activation of these precursors to myofibroblasts requires matrix stiffness, which results in part from increased collagen cross-linking mediated by lysyl oxidase (LOX) family proteins. The aims of this study were to characterize the mechanical changes of early fibrosis, to identify the cells responsible for LOX production in early injury, and to determine which cells in normal liver produce collagens and elastins, which serve as substrates for LOXs early after injury. Hepatocytes and liver nonparenchymal cells were isolated from normal and early-injured liver and examined immediately for expression of LOXs and matrix proteins. We found that stellate cells and portal fibroblasts were the major cellular sources of fibrillar collagens and LOXs in normal liver and early after injury (1 day after bile duct ligation and 2 and 7 days after CCl(4) injury). Activity assays using stellate cells and portal fibroblasts in culture demonstrated significant increases in LOX family enzymatic activity as cells became myofibroblastic. LOX family-mediated deoxypyridinoline and pyridinoline cross-links increased after CCl(4)-mediated injury. There was a significant association between liver stiffness (as quantified by the shear storage modulus G') and deoxypyridinoline levels; increased deoxypyridinoline levels were also coincident with significantly increased elastic resistance to large strain deformations, consistent with increased cross-linking of the extracellular matrix. These data suggest a model in which the liver is primed to respond quickly to injury, activating potential mechanical feed-forward mechanisms.
Figures

References
-
- Barry-Hamilton V, Spangler R, Marshall D, McCauley S, Rodriguez HM, Oyasu M, Mikels A, Vaysberg M, Ghermazien H, Wai C, Garcia CA, Velayo AC, Jorgensen B, Biermann D, Tsai D, Green J, Zaffryar-Eilot S, Holzer A, Ogg S, Thai D, Neufeld G, Van Vlasselaer P, Smith V. Allosteric inhibition of lysyl oxidase-like-2 impedes the development of a pathologic microenvironment. Nat Med 16: 1009–1017, 2010 - PubMed
-
- Bourbonnais E, Raymond VA, Ethier C, Nguyen BN, El-Leil MS, Meloche S, Bilodeau M. Liver fibrosis protects mice from acute hepatocellular injury. Gastroenterology 142: 130–139, 2012 - PubMed
-
- Brenner DA, Waterboer T, Choi SK, Lindquist JN, Stefanovic B, Burchardt E, Yamauchi M, Gillan A, Rippe RA. New aspects of hepatic fibrosis. J Hepatol 32 Suppl: 32–38, 2000 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

