TNF can activate RIPK3 and cause programmed necrosis in the absence of RIPK1
- PMID: 23328672
- PMCID: PMC3563989
- DOI: 10.1038/cddis.2012.201
TNF can activate RIPK3 and cause programmed necrosis in the absence of RIPK1
Abstract
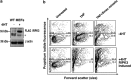
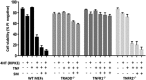
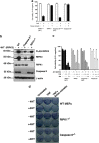
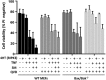
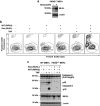
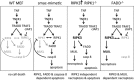
Ligation of tumor necrosis factor receptor 1 (TNFR1) can cause cell death by caspase 8 or receptor-interacting protein kinase 1 (RIPK1)- and RIPK3-dependent mechanisms. It has been assumed that because RIPK1 bears a death domain (DD), but RIPK3 does not, RIPK1 is necessary for recruitment of RIPK3 into signaling and death-inducing complexes. To test this assumption, we expressed elevated levels of RIPK3 in murine embryonic fibroblasts (MEFs) from wild-type (WT) and gene-deleted mice, and exposed them to TNF. Neither treatment with TNF nor overexpression of RIPK3 alone caused MEFs to die, but when levels of RIPK3 were increased, addition of TNF killed WT, Ripk1(-/-), caspase 8(-/-), and Bax(-/-)/Bak(-/-) MEFs, even in the presence of the broad-spectrum caspase inhibitor Q-VD-OPh. In contrast, Tnfr1(-/-) and Tradd(-/-) MEFs did not die. These results show for the first time that in the absence of RIPK1, TNF can activate RIPK3 to induce cell death both by a caspase 8-dependent mechanism and by a separate Bax/Bak- and caspase-independent mechanism. RIPK1 is therefore not essential for TNF to activate RIPK3 to induce necroptosis nor for the formation of a functional ripoptosome/necrosome.
Figures

References
-
- Baud V, Karin M. Signal transduction by tumor necrosis factor and its relatives. Trends Cell Biol. 2001;11:372–377. - PubMed
-
- Laster SM, Wood JG, Gooding LR. Tumor necrosis factor can induce both apoptic and necrotic forms of cell lysis. J Immunol. 1988;141:2629–2634. - PubMed
-
- Beg AA, Baltimore D. An essential role for Nf-Kappa-B in preventing Tnf-alpha-induced cell death. Science. 1996;274:782–784. - PubMed
-
- Chinnaiyan AM, Tepper CG, Seldin MF, Orourke K, Kischkel FC, Hellbardt S, et al. Fadd/mort1 is a common mediator of cd95 (fas/apo-1) and tumor necrosis factor receptor-induced apoptosis. J Biol Chem. 1996;271:4961–4965. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

