Cycles of light and dark co-ordinate reversible colony differentiation in Listeria monocytogenes
- PMID: 23331346
- PMCID: PMC3610012
- DOI: 10.1111/mmi.12140
Cycles of light and dark co-ordinate reversible colony differentiation in Listeria monocytogenes
Abstract
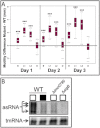
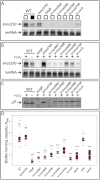
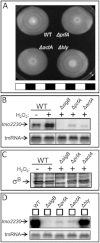
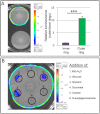
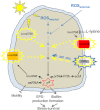
Recently, several light receptors have been identified in non-phototrophic bacteria, but their physiological roles still remain rather elusive. Here we show that colonies of the saprophytic bacterium Listeria monocytogenes undergo synchronized multicellular behaviour on agar plates, in response to oscillating light/dark conditions, giving rise to alternating ring formation (opaque and translucent rings). On agar plates, bacteria from opaque rings survive increased levels of reactive oxygen species (ROS), as well as repeated cycles of light and dark, better than bacteria from translucent rings. The ring formation is strictly dependent on a blue-light receptor, Lmo0799, acting through the stress-sigma factor, σ(B) . A transposon screening identified 48 mutants unable to form rings at alternating light conditions, with several of them showing a decreased σ(B) activity/level. However, some of the tested mutants displayed a varied σ(B) activity depending on which of the two stress conditions tested (light or H(2) O(2) exposure). Intriguingly, the transcriptional regulator PrfA and the virulence factor ActA were shown to be required for ring formation by a mechanism involving activation of σ(B) . All in all, this suggests a distinct pathway for Lmo0799 that converge into a common signalling pathway for σ(B) activation. Our results show that night and day cycles co-ordinate a reversible differentiation of a L. monocytogenes colony at room temperature, by a process synchronized by a blue-light receptor and σ(B) .
© 2013 Blackwell Publishing Ltd.
Figures


Similar articles
-
Blue-Light Inhibition of Listeria monocytogenes Growth Is Mediated by Reactive Oxygen Species and Is Influenced by σB and the Blue-Light Sensor Lmo0799.Appl Environ Microbiol. 2016 Jun 13;82(13):4017-4027. doi: 10.1128/AEM.00685-16. Print 2016 Jul 1. Appl Environ Microbiol. 2016. PMID: 27129969 Free PMC article.
-
Blue and red light modulates SigB-dependent gene transcription, swimming motility and invasiveness in Listeria monocytogenes.PLoS One. 2011 Jan 11;6(1):e16151. doi: 10.1371/journal.pone.0016151. PLoS One. 2011. PMID: 21264304 Free PMC article.
-
Flick of a switch: regulatory mechanisms allowing Listeria monocytogenes to transition from a saprophyte to a killer.Microbiology (Reading). 2019 Aug;165(8):819-833. doi: 10.1099/mic.0.000808. Epub 2019 May 20. Microbiology (Reading). 2019. PMID: 31107205 Review.
-
Mild Stress Conditions during Laboratory Culture Promote the Proliferation of Mutations That Negatively Affect Sigma B Activity in Listeria monocytogenes.J Bacteriol. 2020 Apr 9;202(9):e00751-19. doi: 10.1128/JB.00751-19. Print 2020 Apr 9. J Bacteriol. 2020. PMID: 32094160 Free PMC article.
-
Cross Talk between SigB and PrfA in Listeria monocytogenes Facilitates Transitions between Extra- and Intracellular Environments.Microbiol Mol Biol Rev. 2019 Sep 4;83(4):e00034-19. doi: 10.1128/MMBR.00034-19. Print 2019 Nov 20. Microbiol Mol Biol Rev. 2019. PMID: 31484692 Free PMC article. Review.
Cited by
-
Listeria monocytogenes Requires the RsbX Protein To Prevent SigB Activation under Nonstressed Conditions.J Bacteriol. 2022 Jan 18;204(1):e0048621. doi: 10.1128/JB.00486-21. Epub 2021 Oct 25. J Bacteriol. 2022. PMID: 34694900 Free PMC article.
-
Listeria monocytogenes: survival and adaptation in the gastrointestinal tract.Front Cell Infect Microbiol. 2014 Feb 5;4:9. doi: 10.3389/fcimb.2014.00009. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 24551601 Free PMC article. Review.
-
A Look inside the Listeria monocytogenes Biofilms Extracellular Matrix.Microorganisms. 2016 Jul 5;4(3):22. doi: 10.3390/microorganisms4030022. Microorganisms. 2016. PMID: 27681916 Free PMC article. Review.
-
Light/Dark and Temperature Cycling Modulate Metabolic Electron Flow in Pseudomonas aeruginosa Biofilms.mBio. 2022 Aug 30;13(4):e0140722. doi: 10.1128/mbio.01407-22. Epub 2022 Aug 8. mBio. 2022. PMID: 35938725 Free PMC article.
-
Pathogenicity and virulence of Listeria monocytogenes: A trip from environmental to medical microbiology.Virulence. 2021 Dec;12(1):2509-2545. doi: 10.1080/21505594.2021.1975526. Virulence. 2021. PMID: 34612177 Free PMC article.
References
-
- Aravind L, Koonin EV. The STAS domain – a link between anion transporters and antisigma-factor antagonists. Curr Biol. 2000;10:R53–R55. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

