Host cell factors in filovirus entry: novel players, new insights
- PMID: 23342362
- PMCID: PMC3528269
- DOI: 10.3390/v4123336
Host cell factors in filovirus entry: novel players, new insights
Abstract
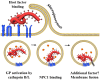
Filoviruses cause severe hemorrhagic fever in humans with high case-fatality rates. The cellular factors exploited by filoviruses for their spread constitute potential targets for intervention, but are incompletely defined. The viral glycoprotein (GP) mediates filovirus entry into host cells. Recent studies revealed important insights into the host cell molecules engaged by GP for cellular entry. The binding of GP to cellular lectins was found to concentrate virions onto susceptible cells and might contribute to the early and sustained infection of macrophages and dendritic cells, important viral targets. Tyrosine kinase receptors were shown to promote macropinocytic uptake of filoviruses into a subset of susceptible cells without binding to GP, while interactions between GP and human T cell Ig mucin 1 (TIM-1) might contribute to filovirus infection of mucosal epithelial cells. Moreover, GP engagement of the cholesterol transporter Niemann-Pick C1 was demonstrated to be essential for GP-mediated fusion of the viral envelope with a host cell membrane. Finally, mutagenic and structural analyses defined GP domains which interact with these host cell factors. Here, we will review the recent progress in elucidating the molecular interactions underlying filovirus entry and discuss their implications for our understanding of the viral cell tropism.
Figures

References
-
- Kuhn J.H., Becker S., Ebihara H., Geisbert T.W., Jahrling P.B., Kawaoka Y., Netesov S.V., Nichol S.T., Peters C.J., Volchkov V.E., et al. Family Filoviridae. In: King A.M.Q., Adams M.J., Carstens E.B., Lefkowitz E.J., editors. Virus Taxonomy—Ninth Report of the International Committee on Taxonomy of Viruses. Elsevier/Academic Press; London, UK: 2001. pp. 665–671.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
