Early postnatal development of GABAergic presynaptic inhibition of Ia proprioceptive afferent connections in mouse spinal cord
- PMID: 23343895
- PMCID: PMC3628030
- DOI: 10.1152/jn.00783.2012
Early postnatal development of GABAergic presynaptic inhibition of Ia proprioceptive afferent connections in mouse spinal cord
Abstract
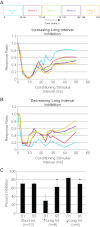
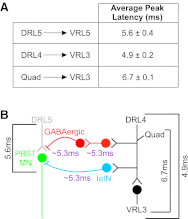
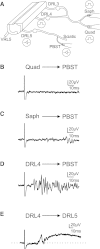
Sensory feedback is critical for normal locomotion and adaptation to external perturbations during movement. Feedback provided by group Ia afferents influences motor output both directly through monosynaptic connections and indirectly through spinal interneuronal circuits. For example, the circuit responsible for reciprocal inhibition, which acts to prevent co-contraction of antagonist flexor and extensor muscles, is driven by Ia afferent feedback. Additionally, circuits mediating presynaptic inhibition can limit Ia afferent synaptic transmission onto central neuronal targets in a task-specific manner. These circuits can also be activated by stimulation of proprioceptive afferents. Rodent locomotion rapidly matures during postnatal development; therefore, we assayed the functional status of reciprocal and presynaptic inhibitory circuits of mice at birth and compared responses with observations made after 1 wk of postnatal development. Using extracellular physiological techniques from isolated and hemisected spinal cord preparations, we demonstrate that Ia afferent-evoked reciprocal inhibition is as effective at blocking antagonist motor neuron activation at birth as at 1 wk postnatally. In contrast, at birth conditioning stimulation of muscle nerve afferents failed to evoke presynaptic inhibition sufficient to block functional transmission at synapses between Ia afferents and motor neurons, even though dorsal root potentials could be evoked by stimulating the neighboring dorsal root. Presynaptic inhibition at this synapse was readily observed, however, at the end of the first postnatal week. These results indicate Ia afferent feedback from the periphery to central spinal circuits is only weakly gated at birth, which may provide enhanced sensitivity to peripheral feedback during early postnatal experiences.
Figures


References
-
- Alvarez FJ. Anatomical basis for presynaptic inhibition of primary sensory fibers. In: Presynaptic Inhibition and Neural Control, edited by Rudomin P, Romo R, Mendell L. New York: Oxford University Press, 1998, p. 13–49
-
- Arber S, Ladle DR, Lin JH, Frank E, Jessell TM. ETS gene Er81 controls the formation of functional connections between group Ia sensory afferents and motor neurons. Cell 101: 485–498, 2000 - PubMed
-
- Baldissera F, Hultborn H, Illert M. Integration in spinal neuronal systems. In: Handbook of Physiology. The Nervous System. Bethesda, MD: Am. Physiol. Soc., 1981, p. 509–596
-
- Banks RW. The muscle spindle. In: Peripheral Neuropathy, edited by Dyck PJ, Thomas PK. Philadelphia, PA: Saunders, 2005, p. 131–150
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

