Pip6-PMO, A New Generation of Peptide-oligonucleotide Conjugates With Improved Cardiac Exon Skipping Activity for DMD Treatment
- PMID: 23344180
- PMCID: PMC3438601
- DOI: 10.1038/mtna.2012.30
Pip6-PMO, A New Generation of Peptide-oligonucleotide Conjugates With Improved Cardiac Exon Skipping Activity for DMD Treatment
Abstract
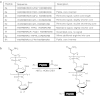
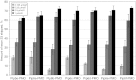
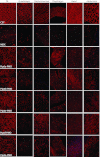
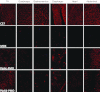
Antisense oligonucleotides (AOs) are currently the most promising therapeutic intervention for Duchenne muscular dystrophy (DMD). AOs modulate dystrophin pre-mRNA splicing, thereby specifically restoring the dystrophin reading frame and generating a truncated but semifunctional dystrophin protein. Challenges in the development of this approach are the relatively poor systemic AO delivery and inefficient dystrophin correction in affected non-skeletal muscle tissues, including the heart. We have previously reported impressive heart activity including high-splicing efficiency and dystrophin restoration following a single administration of an arginine-rich cell-penetrating peptide (CPPs) conjugated to a phosphorodiamidate morpholino oligonucleotide (PMO): Pip5e-PMO. However, the mechanisms underlying this activity are poorly understood. Here, we report studies involving single dose administration (12.5 mg/kg) of derivatives of Pip5e-PMO, consecutively assigned as Pip6-PMOs. These peptide-PMOs comprise alterations to the central hydrophobic core of the Pip5e peptide and illustrate that certain changes to the peptide sequence improves its activity; however, partial deletions within the hydrophobic core abolish its efficiency. Our data indicate that the hydrophobic core of the Pip sequences is critical for PMO delivery to the heart and that specific modifications to this region can enhance activity further. The results have implications for therapeutic PMO development for DMD.
Figures


References
-
- Emery AE. Population frequencies of inherited neuromuscular diseases–a world survey. Neuromuscul Disord. 1991;1:19–29. - PubMed
-
- Wilton SD, Lloyd F, Carville K, Fletcher S, Honeyman K, Agrawal S.et al. (1999Specific removal of the nonsense mutation from the mdx dystrophin mRNA using antisense oligonucleotides Neuromuscul Disord 9330–338. - PubMed
-
- Dunckley MG, Manoharan M, Villiet P, Eperon IC., and, Dickson G. Modification of splicing in the dystrophin gene in cultured Mdx muscle cells by antisense oligoribonucleotides. Hum Mol Genet. 1998;7:1083–1090. - PubMed
-
- Dickson G, Hill V., and, Graham IR. Screening for antisense modulation of dystrophin pre-mRNA splicing. Neuromuscul Disord. 2002;12 Suppl 1:S67–S70. - PubMed
-
- Alter J, Lou F, Rabinowitz A, Yin H, Rosenfeld J, Wilton SD.et al. (2006Systemic delivery of morpholino oligonucleotide restores dystrophin expression bodywide and improves dystrophic pathology Nat Med 12175–177. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

