Ca2+ signals generated by CatSper and Ca2+ stores regulate different behaviors in human sperm
- PMID: 23344959
- PMCID: PMC3585060
- DOI: 10.1074/jbc.M112.439356
Ca2+ signals generated by CatSper and Ca2+ stores regulate different behaviors in human sperm
Abstract
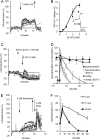
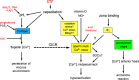
[Ca(2+)]i signaling regulates sperm motility, enabling switching between functionally different behaviors that the sperm must employ as it ascends the female tract and fertilizes the oocyte. We report that different behaviors in human sperm are recruited according to the Ca(2+) signaling pathway used. Activation of CatSper (by raising pHi or stimulating with progesterone) caused sustained [Ca(2+)]i elevation but did not induce hyperactivation, the whiplash-like behavior required for progression along the oviduct and penetration of the zona pellucida. In contrast, penetration into methylcellulose (mimicking penetration into cervical mucus or cumulus matrix) was enhanced by activation of CatSper. NNC55-0396, which abolishes CatSper currents in human sperm, inhibited this effect. Treatment with 5 μm thimerosal to mobilize stored Ca(2+) caused sustained [Ca(2+)]i elevation and induced strong, sustained hyperactivation that was completely insensitive to NNC55-0396. Thimerosal had no effect on penetration into methylcellulose. 4-Aminopyridine, a powerful modulator of sperm motility, both raised pHi and mobilized Ca(2+) stored in sperm (and from microsomal membrane preparations). 4-Aminopyridine-induced hyperactivation even in cells suspended in Ca(2+)-depleted medium and also potentiated penetration into methylcellulose. The latter effect was sensitive to NNC55-039, but induction of hyperactivation was not. We conclude that these two components of the [Ca(2+)]i signaling apparatus have strikingly different effects on sperm motility. Furthermore, since stored Ca(2+) at the sperm neck can be mobilized by Ca(2+)-induced Ca(2+) release, we propose that CatSper activation can elicit functionally different behaviors according to the sensitivity of the Ca(2+) store, which may be regulated by capacitation and NO from the cumulus.
Figures



References
-
- Stauss C. R., Votta T. J., Suarez S. S. (1995) Sperm motility hyperactivation facilitates penetration of the hamster zona pellucida. Biol. Reprod. 53, 1280–1285 - PubMed
-
- Suarez S. S. (2008) Control of hyperactivation in sperm. Hum. Reprod. Update 14, 647–657 - PubMed
-
- Yanagimachi R. (1970) The movement of golden hamster spermatozoa before and after capacitation. J. Reprod. Fertil. 23, 193–196 - PubMed
-
- Drobnis E. Z., Yudin A. I., Cherr G. N., Katz D. F. (1988) Hamster sperm penetration of the zona pellucida. Kinematic analysis and mechanical implications. Dev. Biol. 130, 311–323 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

