Deletion of the NMDA-NR1 receptor subunit gene in the mouse nucleus accumbens attenuates apomorphine-induced dopamine D1 receptor trafficking and acoustic startle behavior
- PMID: 23345061
- PMCID: PMC3894854
- DOI: 10.1002/syn.21637
Deletion of the NMDA-NR1 receptor subunit gene in the mouse nucleus accumbens attenuates apomorphine-induced dopamine D1 receptor trafficking and acoustic startle behavior
Abstract
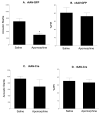
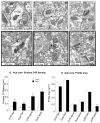
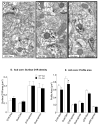
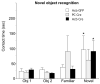
The nucleus accumbens (Acb) contains subpopulations of neurons defined by their receptor content and potential involvement in sensorimotor gating and other behaviors that are dysfunctional in schizophrenia. In Acb neurons, the NMDA NR1 (NR1) subunit is coexpressed not only with the dopamine D1 receptor (D1R), but also with the µ-opioid receptor (µ-OR), which mediates certain behaviors that are adversely impacted by schizophrenia. The NMDA-NR1 subunit has been suggested to play a role in the D1R trafficking and behavioral dysfunctions resulting from systemic administration of apomorphine, a D1R and dopamine D2 receptor agonist that impacts prepulse inhibition to auditory-evoked startle (AS). Together, this evidence suggests that the NMDA receptor may regulate D1R trafficking in Acb neurons, including those expressing µ-OR, in animals exposed to auditory startle and apomorphine. We tested this hypothesis by combining spatial-temporal gene deletion technology, dual labeling immunocytochemistry, and behavioral analysis. Deleting NR1 in Acb neurons prevented the increase in the dendritic density of plasma membrane D1Rs in single D1R and dual (D1R and µ-OR) labeled dendrites in the Acb in response to apomorphine and AS. Deleting NR1 also attenuated the decrease in AS induced by apomorphine. In the absence of apomorphine and startle, deletion of Acb NR1 diminished social interaction, without affecting novel object recognition, or open field activity. These results suggest that NR1 expression in the Acb is essential for apomorphine-induced D1R surface trafficking, as well as auditory startle and social behaviors that are impaired in multiple psychiatric disorders.
Copyright © 2013 Wiley Periodicals, Inc.
Figures

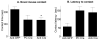
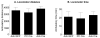
Similar articles
-
Preferential relocation of the N-methyl-D-aspartate receptor NR1 subunit in nucleus accumbens neurons that contain dopamine D1 receptors in rats showing an apomorphine-induced sensorimotor gating deficit.Neuroscience. 2008 Jun 26;154(3):965-77. doi: 10.1016/j.neuroscience.2008.04.009. Epub 2008 Apr 12. Neuroscience. 2008. PMID: 18479834 Free PMC article.
-
Dendritic distributions of dopamine D1 receptors in the rat nucleus accumbens are synergistically affected by startle-evoking auditory stimulation and apomorphine.Neuroscience. 2007 Jun 8;146(4):1593-605. doi: 10.1016/j.neuroscience.2007.04.005. Epub 2007 May 9. Neuroscience. 2007. PMID: 17490822 Free PMC article.
-
Overlapping intracellular and differential synaptic distributions of dopamine D1 and glutamate N-methyl-D-aspartate receptors in rat nucleus accumbens.J Comp Neurol. 2005 Nov 28;492(4):442-55. doi: 10.1002/cne.20740. J Comp Neurol. 2005. PMID: 16228995 Free PMC article.
-
The role of functional postsynaptic NMDA receptors in the central nucleus of the amygdala in opioid dependence.Vitam Horm. 2010;82:145-66. doi: 10.1016/S0083-6729(10)82008-4. Vitam Horm. 2010. PMID: 20472137 Free PMC article. Review.
-
The NMDA/D1 receptor complex as a new target in drug development.Curr Top Med Chem. 2006;6(8):801-8. doi: 10.2174/156802606777057562. Curr Top Med Chem. 2006. PMID: 16719818 Review.
Cited by
-
Canonical and Non-Canonical Antipsychotics' Dopamine-Related Mechanisms of Present and Next Generation Molecules: A Systematic Review on Translational Highlights for Treatment Response and Treatment-Resistant Schizophrenia.Int J Mol Sci. 2023 Mar 21;24(6):5945. doi: 10.3390/ijms24065945. Int J Mol Sci. 2023. PMID: 36983018 Free PMC article.
-
The involvement of N-methyl-D-aspartate receptor (NMDAR) subunit NR1 in the pathophysiology of schizophrenia.Acta Biochim Biophys Sin (Shanghai). 2016 Mar;48(3):209-19. doi: 10.1093/abbs/gmv135. Epub 2016 Feb 1. Acta Biochim Biophys Sin (Shanghai). 2016. PMID: 26837414 Free PMC article. Review.
-
Hypothesizing dopaminergic genetic antecedents in schizophrenia and substance seeking behavior.Med Hypotheses. 2014 May;82(5):606-14. doi: 10.1016/j.mehy.2014.02.019. Epub 2014 Feb 26. Med Hypotheses. 2014. PMID: 24636783 Free PMC article.
-
Sociability development in mice with cell-specific deletion of the NMDA receptor NR1 subunit gene.Genes Brain Behav. 2020 Jan;19(1):e12624. doi: 10.1111/gbb.12624. Epub 2019 Dec 12. Genes Brain Behav. 2020. PMID: 31721416 Free PMC article.
-
Sex differences in NMDA GluN1 plasticity in rostral ventrolateral medulla neurons containing corticotropin-releasing factor type 1 receptor following slow-pressor angiotensin II hypertension.Neuroscience. 2015 Oct 29;307:83-97. doi: 10.1016/j.neuroscience.2015.08.029. Epub 2015 Aug 22. Neuroscience. 2015. PMID: 26306872 Free PMC article.
References
-
- Bermak JC, Li M, Bullock C, Zhou QY. Regulation of transport of the dopamine D1 receptor by a new membrane-associated ER protein. Nat. Cell Biol. 2001;3:492–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
