Regulation of integrin αV subunit expression by sulfatide in hepatocellular carcinoma cells
- PMID: 23345412
- PMCID: PMC3606000
- DOI: 10.1194/jlr.M031450
Regulation of integrin αV subunit expression by sulfatide in hepatocellular carcinoma cells
Abstract
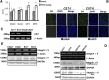
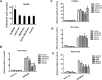
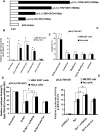
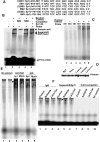
Integrin is important in migration and metastasis of tumor cells. Changes of integrin expression and distribution will cause an alteration of cellular adhesion and migration behaviors. In this study, we investigated sulfatide regulation of the integrin αV subunit expression in hepatoma cells and observed that either exogenous or endogenous sulfatide elicited a robust upregulation of integrin αV subunit mRNA and protein expression in hepatoma cells. This regulatory effect occurred with a corresponding phosphorylation (T739) of the transcription factor Sp1. Based on the electrophoretic mobility shift assay, sulfatide enhanced the integrin αV promoter activity and strengthened the Sp1 complex super-shift. The results of chromatin immunoprecipitation analysis also indicated that sulfatide enhanced Sp1 binding to the integrin αV promoter in vivo. Silence of Sp1 diminished the stimulation of integrin αV expression by sulfatide. In the early stage of sulfatide stimulation, phosphorylation of Erk as well as c-Src was noted, and inhibition of Erk activation with either U0126 or PD98059 significantly suppressed Sp1 phosphorylation and integrin αV expression. We demonstrated that sulfatide regulated integrin αV expression and cell adhesion, which was associated with Erk activation.
Figures




Similar articles
-
Sulfatide interacts with and activates integrin αVβ3 in human hepatocellular carcinoma cells.Oncotarget. 2016 Jun 14;7(24):36563-36576. doi: 10.18632/oncotarget.9095. Oncotarget. 2016. PMID: 27145276 Free PMC article.
-
SIN3B promotes integrin αV subunit gene transcription and cell migration of hepatocellular carcinoma.J Mol Cell Biol. 2019 May 1;11(5):421-432. doi: 10.1093/jmcb/mjy050. J Mol Cell Biol. 2019. PMID: 30215728 Free PMC article.
-
BRD1-Mediated Acetylation Promotes Integrin αV Gene Expression Via Interaction with Sulfatide.Mol Cancer Res. 2018 Apr;16(4):610-622. doi: 10.1158/1541-7786.MCR-17-0527. Epub 2018 Feb 16. Mol Cancer Res. 2018. PMID: 29453316
-
Sulfatide epigenetically regulates miR-223 and promotes the migration of human hepatocellular carcinoma cells.J Hepatol. 2014 Apr;60(4):792-801. doi: 10.1016/j.jhep.2013.12.004. Epub 2013 Dec 11. J Hepatol. 2014. PMID: 24333181
-
An αv-RGD Integrin Inhibitor Toolbox: Drug Discovery Insight, Challenges and Opportunities.Angew Chem Int Ed Engl. 2018 Mar 19;57(13):3298-3321. doi: 10.1002/anie.201707948. Epub 2018 Feb 21. Angew Chem Int Ed Engl. 2018. PMID: 28944552 Review.
Cited by
-
Vimentin is important in the neural differentiation of PC12 cells promoted by sialylation.Glycoconj J. 2017 Feb;34(1):51-59. doi: 10.1007/s10719-016-9727-6. Epub 2016 Oct 10. Glycoconj J. 2017. PMID: 27726058
-
Prognostic and immunological role of sulfatide-related lncRNAs in hepatocellular carcinoma.Front Oncol. 2023 Feb 1;13:1091132. doi: 10.3389/fonc.2023.1091132. eCollection 2023. Front Oncol. 2023. PMID: 36816914 Free PMC article.
-
LPPR4 promotes peritoneal metastasis via Sp1/integrin α/FAK signaling in gastric cancer.Am J Cancer Res. 2020 Mar 1;10(3):1026-1044. eCollection 2020. Am J Cancer Res. 2020. PMID: 32266108 Free PMC article.
-
Integrin αV modulates the cellular pharmacology of copper and cisplatin by regulating expression of the influx transporter CTR1.Oncoscience. 2014 Mar 24;1(3):185-195. doi: 10.18632/oncoscience.22. eCollection 2014. Oncoscience. 2014. PMID: 25594011 Free PMC article.
-
Sulfatide interacts with and activates integrin αVβ3 in human hepatocellular carcinoma cells.Oncotarget. 2016 Jun 14;7(24):36563-36576. doi: 10.18632/oncotarget.9095. Oncotarget. 2016. PMID: 27145276 Free PMC article.
References
-
- Schwartz M. A., Assoian R. K. 2001. Integrins and cell proliferation: regulation of cyclin-dependent kinases via cytoplasmic signaling pathways. J. Cell Sci. 114: 2553–2560. - PubMed
-
- Shimaoka M., Takagi J., Springer T. A. 2002. Conformational regulation of integrin structure and function. Annu. Rev. Biophys. Biomol. Struct. 31: 485–516. - PubMed
-
- Zahir N., Weaver V. M. 2004. Death in the third dimension: apoptosis regulation and tissue architecture. Curr. Opin. Genet. Dev. 14: 71–80. - PubMed
-
- Gao B., Saba T. M., Tsan M. F. 2002. Role of alpha(v)beta(3)-integrin in TNF-alpha-induced endothelial cell migration. Am. J. Physiol. Cell Physiol. 283: C1196–C1205. - PubMed
-
- Li H., Ge C., Zhao F., Yan M., Hu C., Jia D., Tian H., Zhu M., Chen T., Jiang G., et al. 2011. Hypoxia-inducible factor 1 alpha-activated angiopoietin-like protein 4 contributes to tumor metastasis via vascular cell adhesion molecule-1/integrin beta1 signaling in human hepatocellular carcinoma. Hepatology. 54: 910–919. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

