Plasticity within the αβ⁺CD4⁺ T-cell lineage: when, how and what for?
- PMID: 23345540
- PMCID: PMC3603458
- DOI: 10.1098/rsob.120157
Plasticity within the αβ⁺CD4⁺ T-cell lineage: when, how and what for?
Abstract
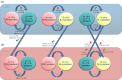
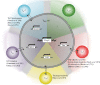
Following thymic output, αβ⁺CD4⁺ T cells become activated in the periphery when they encounter peptide-major histocompatibility complex. A combination of cytokine and co-stimulatory signals instructs the differentiation of T cells into various lineages and subsequent expansion and contraction during an appropriate and protective immune response. Our understanding of the events leading to T-cell lineage commitment has been dominated by a single fate model describing the commitment of T cells to one of several helper (T(H)), follicular helper (T(FH)) or regulatory (T(REG)) phenotypes. Although a single lineage-committed and dedicated T cell may best execute a single function, the view of a single fate for T cells has recently been challenged. A relatively new paradigm in αβ⁺CD4⁺ T-cell biology indicates that T cells are much more flexible than previously appreciated, with the ability to change between helper phenotypes, between helper and follicular helper, or, most extremely, between helper and regulatory functions. In this review, we comprehensively summarize the recent literature identifying when T(H) or T(REG) cell plasticity occurs, provide potential mechanisms of plasticity and ask if T-cell plasticity is beneficial or detrimental to immunity.
Figures


References
-
- Gerriets VA, Rathmell JC. 2012. Metabolic pathways in T cell fate and function. Trends Immunol. 33, 168–173 10.1016/j.it.2012.01.010 (doi:10.1016/j.it.2012.01.010) - DOI - PMC - PubMed
-
- Okoye IS, Wilson MS. 2011. CD4+ T helper 2 cells: microbial triggers, differentiation requirements and effector functions. Immunology 134, 368–377 10.1111/j.1365-2567.2011.03497.x (doi:10.1111/j.1365-2567.2011.03497.x) - DOI - PMC - PubMed
-
- Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. 1995. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 155, 1151–1164 - PubMed
-
- Bluestone JA, Abbas AK. 2003. Natural versus adaptive regulatory T cells. Nat. Rev. Immunol. 3, 253–257 10.1038/nri1032 (doi:10.1038/nri1032) - DOI - PubMed
-
- Chen Y, Kuchroo V, Inobe J, Hafler D, Weiner H. 1994. Regulatory T cell clones induced by oral tolerance: suppression of autoimmune encephalomyelitis. Science 265, 1237–1240 10.1126/science.7520605 (doi:10.1126/science.7520605) - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
