A multi-scale modeling framework for individualized, spatiotemporal prediction of drug effects and toxicological risk
- PMID: 23346056
- PMCID: PMC3551257
- DOI: 10.3389/fphar.2012.00204
A multi-scale modeling framework for individualized, spatiotemporal prediction of drug effects and toxicological risk
Abstract
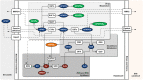
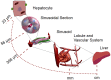
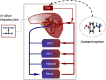
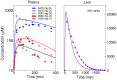
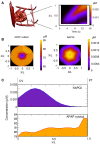
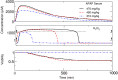
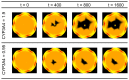
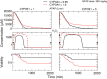
In this study, we focus on a novel multi-scale modeling approach for spatiotemporal prediction of the distribution of substances and resulting hepatotoxicity by combining cellular models, a 2D liver model, and whole body model. As a case study, we focused on predicting human hepatotoxicity upon treatment with acetaminophen based on in vitro toxicity data and potential inter-individual variability in gene expression and enzyme activities. By aggregating mechanistic, genome-based in silico cells to a novel 2D liver model and eventually to a whole body model, we predicted pharmacokinetic properties, metabolism, and the onset of hepatotoxicity in an in silico patient. Depending on the concentration of acetaminophen in the liver and the accumulation of toxic metabolites, cell integrity in the liver as a function of space and time as well as changes in the elimination rate of substances were estimated. We show that the variations in elimination rates also influence the distribution of acetaminophen and its metabolites in the whole body. Our results are in agreement with experimental results. What is more, the integrated model also predicted variations in drug toxicity depending on alterations of metabolic enzyme activities. Variations in enzyme activity, in turn, reflect genetic characteristics or diseases of individuals. In conclusion, this framework presents an important basis for efficiently integrating inter-individual variability data into models, paving the way for personalized or stratified predictions of drug toxicity and efficacy.
Keywords: acetaminophen; drug metabolism; hepatotoxicity; pharmacokinetics; toxicity testing.
Figures


References
-
- Adjei A. A., Gaedigk A., Simon S. D., Weinshilboum R. M., Leeder J. S. (2008). Interindividual variability in acetaminophen sulfation by human fetal liver: implications for pharmacogenetic investigations of drug-induced birth defects. Birth Defects Res. Part A Clin. Mol. Teratol. 82, 155–16510.1002/bdra.20535 - DOI - PubMed
-
- Arias I. M., Alter H. J., Boyer J. L., Cohen D. E., Fausto N., Shaftitz D. A., et al. (2009). The Liver: Biology and Pathobiology. Australia: John Wiley and Sons
-
- Basketter D. A., Clewell H., Kimber I., Rossi A., Blaauboer B., Burrier R., et al. (2012). A roadmap for the development of alternative (non-animal) methods for systemic toxicity testing – t4 report*. ALTEX 29, 3–91 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

