Bone circulatory disturbances in the development of spontaneous bacterial chondronecrosis with osteomyelitis: a translational model for the pathogenesis of femoral head necrosis
- PMID: 23346077
- PMCID: PMC3550519
- DOI: 10.3389/fendo.2012.00183
Bone circulatory disturbances in the development of spontaneous bacterial chondronecrosis with osteomyelitis: a translational model for the pathogenesis of femoral head necrosis
Abstract
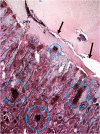
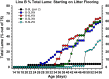
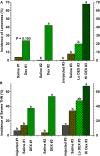
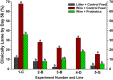
This review provides a comprehensive overview of the vascularization of the avian growth plate and its subsequent role in the pathogenesis of bacterial chondronecrosis with osteomyelitis (BCO, femoral head necrosis). BCO sporadically causes high incidences of lameness in rapidly growing broiler (meat-type) chickens. BCO is believed to be initiated by micro-trauma to poorly mineralized columns of cartilage cells in the proximal growth plates of the leg bones, followed by colonization by hematogenously distributed opportunistic bacteria. Inadequate blood flow to the growth plate, vascular occlusion, and structural limitations of the microvasculature all have been implicated in the pathogenesis of BCO. Treatment strategies have been difficult to investigate because under normal conditions the incidence of BCO typically is low and sporadic. Rearing broilers on wire flooring triggers the spontaneous development of high incidences of lameness attributable to pathognomonic BCO lesions. Wire flooring imposes persistent footing instability and is thought to accelerate the development of BCO by amplifying the torque and shear stress imposed on susceptible leg joints. Wire flooring per se also constitutes a significant chronic stressor that promotes bacterial proliferation attributed to stress-mediated immunosuppression. Indeed, dexamethasone-mediated immunosuppression causes broilers to develop lameness primarily associated with avascular necrosis and BCO. Prophylactic probiotic administration consistently reduces the incidence of lameness in broilers reared on wire flooring, presumably by reducing bacterial translocation from the gastrointestinal tract that likely contributes to hematogenous infection of the leg bones. The pathogenesis of BCO in broilers is directly relevant to osteomyelitis in growing children, as well as to avascular femoral head necrosis in adults. Our new model for reliably triggering spontaneous osteomyelitis in large numbers of animals represents an important opportunity to conduct translational research focused on developing effective prophylactic and therapeutic treatments.
Keywords: femoral head necrosis; osteomyelitis.
Figures

Similar articles
-
A wire-flooring model for inducing lameness in broilers: evaluation of probiotics as a prophylactic treatment.Poult Sci. 2012 Apr;91(4):870-83. doi: 10.3382/ps.2011-01907. Poult Sci. 2012. PMID: 22399726
-
Bacterial chondronecrosis with osteomyelitis and lameness in broilers: a review.Poult Sci. 2016 Feb;95(2):325-44. doi: 10.3382/ps/pev320. Epub 2015 Nov 2. Poult Sci. 2016. PMID: 26527707 Review.
-
Prophylactic administration of a combined prebiotic and probiotic, or therapeutic administration of enrofloxacin, to reduce the incidence of bacterial chondronecrosis with osteomyelitis in broilers.Poult Sci. 2015 Jan;94(1):25-36. doi: 10.3382/ps/peu025. Poult Sci. 2015. PMID: 25577793
-
Inducing experimental bacterial chondronecrosis with osteomyelitis lameness in broiler chickens using aerosol transmission model.Poult Sci. 2024 Mar;103(3):103460. doi: 10.1016/j.psj.2024.103460. Epub 2024 Jan 14. Poult Sci. 2024. PMID: 38301493 Free PMC article.
-
A Review on Pathophysiology, and Molecular Mechanisms of Bacterial Chondronecrosis and Osteomyelitis in Commercial Broilers.Biomolecules. 2023 Jun 23;13(7):1032. doi: 10.3390/biom13071032. Biomolecules. 2023. PMID: 37509068 Free PMC article. Review.
Cited by
-
Broiler stress responses to light intensity, flooring type, and leg weakness as assessed by heterophil-to-lymphocyte ratios, serum corticosterone, infrared thermography, and latency to lie.Poult Sci. 2020 Jul;99(7):3301-3311. doi: 10.1016/j.psj.2020.03.028. Epub 2020 Apr 15. Poult Sci. 2020. PMID: 32616223 Free PMC article.
-
Effect of Bacillus-direct-fed microbial on leaky gut, serum peptide YY concentration, bone mineralization, and ammonia excretion in neonatal female turkey poults fed with a rye-based diet.Poult Sci. 2020 Sep;99(9):4514-4520. doi: 10.1016/j.psj.2020.06.018. Epub 2020 Jun 30. Poult Sci. 2020. PMID: 32867995 Free PMC article.
-
Utilization of rye as energy source affects bacterial translocation, intestinal viscosity, microbiota composition, and bone mineralization in broiler chickens.Front Genet. 2014 Sep 25;5:339. doi: 10.3389/fgene.2014.00339. eCollection 2014. Front Genet. 2014. PMID: 25309584 Free PMC article.
-
Staphylococcus aureus in Agriculture: Lessons in Evolution from a Multispecies Pathogen.Clin Microbiol Rev. 2021 Feb 10;34(2):e00182-20. doi: 10.1128/CMR.00182-20. Print 2021 Mar 17. Clin Microbiol Rev. 2021. PMID: 33568553 Free PMC article. Review.
-
Effect of endoplasmic reticulum stress on bone and cartilage growth via endochondral ossification in broilers.J Anim Sci. 2025 Jan 4;103:skaf147. doi: 10.1093/jas/skaf147. J Anim Sci. 2025. PMID: 40331700
References
-
- Alderson M., Speers D., Emslie K., Nade S. (1986b). Acute haematogenous osteomyelitis and septic arthritis – a single disease. J. Bone Joint Surg. Br. 68B, 268–274 - PubMed
-
- Alderson M., Nade S. (1987). Natural history of acute septic arthritis in an avian model. J. Orthop. Res. 5, 261–274 - PubMed
-
- Ali S. Y. (1992). “Matrix formation and mineralisation in bone,” in Bone Biology and Skeletal Disorders in Poultry, ed. Whitehead C. C. (Abingdon: Carfax Publishing Company; ), 19–38
LinkOut - more resources
Full Text Sources
Other Literature Sources

