A novel peptide nanomedicine for treatment of pancreatogenic diabetes
- PMID: 23347897
- PMCID: PMC3664121
- DOI: 10.1016/j.nano.2012.12.005
A novel peptide nanomedicine for treatment of pancreatogenic diabetes
Abstract
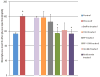
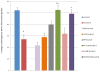
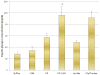
Pancreatogenic diabetes (PD) is a potentially fatal disease that occurs secondary to pancreatic disorders. The current anti-diabetic therapy for PD is fraught with adverse effects that can increase morbidity. Here we investigated the efficacy of novel peptide nanomedicine: pancreatic polypeptide (PP) in sterically stabilized micelles (SSM) for management of PD. PP exhibits significant anti-diabetic efficacy but its short plasma half-life curtails its therapeutic application. To prolong and improve activity of PP in vivo, we evaluated the delivery of PP in SSM. PP-SSM administered to rats with PD, significantly improved glucose tolerance, insulin sensitivity and hepatic glycogen content compared to peptide in buffer. The studies established the importance of micellar nanocarriers in protecting enzyme-labile peptides in vivo and delivering them to target site, thereby enhancing their therapeutic efficacy. In summary, this study demonstrated that PP-SSM is a promising novel anti-diabetic nanomedicine and therefore should be further developed for management of PD.
From the clinical editor: Pancreatic peptide was earlier demonstrated to address pancreatogenic diabetes, but its short half-life represented major difficulties in further development for therapeutic use. PP-SSM (pancreatic polypeptide in sterically stabilized micelles) is a promising novel anti-diabetic nanomedicine that enables prolonged half-life and increased bioactivity of PP, as shown in this novel study, paving the way toward clinical studies in the near future.
Keywords: 1,2-Distearoyl-sn-glycero-3-phosphatidylethanolamine-N-[methoxy (polyethyleneglycol)-2000]; CP; Chronic pancreatitis; DPSPE-PEG(2000); EPR; Enhanced permeation and retention; GLP-1; GLUT4; GTT; Glucagon-like peptide -1; Glucose tolerance test; Glucose transporter 4; IST; Insulin sensitivity test; PBS; PD; PP; Pancreatic polypeptide; Pancreatogenic diabetes; Phosphate buffered saline; SEM; SSM; Standard error of mean; Sterically stabilized micelles; T3cDM; Type 3c diabetes mellitus; VIP; Vasoactive intestinal peptide.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Vasoactive Intestinal Peptide Nanomedicine for the Management of Inflammatory Bowel Disease.Mol Pharm. 2017 Nov 6;14(11):3698-3708. doi: 10.1021/acs.molpharmaceut.7b00452. Epub 2017 Oct 19. Mol Pharm. 2017. PMID: 28991483 Free PMC article.
-
Human pancreatic polypeptide in a phospholipid-based micellar formulation.Pharm Res. 2012 Jun;29(6):1698-711. doi: 10.1007/s11095-012-0718-4. Epub 2012 Mar 8. Pharm Res. 2012. PMID: 22399387 Free PMC article.
-
Micellar Nanomedicine of Novel Fatty Acid Modified Xenopus Glucagon-like Peptide-1: Improved Physicochemical Characteristics and Therapeutic Utilities for Type 2 Diabetes.Mol Pharm. 2017 Nov 6;14(11):3954-3967. doi: 10.1021/acs.molpharmaceut.7b00632. Epub 2017 Oct 6. Mol Pharm. 2017. PMID: 28945431
-
Pancreatogenic diabetes: special considerations for management.Pancreatology. 2011;11(3):279-94. doi: 10.1159/000329188. Epub 2011 Jul 9. Pancreatology. 2011. PMID: 21757968 Review.
-
Pancreatogenic diabetes after pancreatic resection.Pancreatology. 2011;11(2):268-76. doi: 10.1159/000328785. Epub 2011 Jul 5. Pancreatology. 2011. PMID: 21734430 Review.
Cited by
-
Chronic Pancreatitis and Diabetes Mellitus.Curr Treat Options Gastroenterol. 2015 Sep;13(3):319-31. doi: 10.1007/s11938-015-0055-x. Curr Treat Options Gastroenterol. 2015. PMID: 26084759
-
Vasoactive Intestinal Peptide Nanomedicine for the Management of Inflammatory Bowel Disease.Mol Pharm. 2017 Nov 6;14(11):3698-3708. doi: 10.1021/acs.molpharmaceut.7b00452. Epub 2017 Oct 19. Mol Pharm. 2017. PMID: 28991483 Free PMC article.
-
Management of pancreatogenic diabetes: challenges and solutions.Diabetes Metab Syndr Obes. 2016 Aug 25;9:311-5. doi: 10.2147/DMSO.S99701. eCollection 2016. Diabetes Metab Syndr Obes. 2016. PMID: 27601927 Free PMC article. Review.
-
Mechanisms of Post-Pancreatitis Diabetes Mellitus and Cystic Fibrosis-Related Diabetes: A Review of Preclinical Studies.Front Endocrinol (Lausanne). 2021 Sep 10;12:715043. doi: 10.3389/fendo.2021.715043. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34566890 Free PMC article. Review.
-
When Is It Important to Measure Unbound Drug in Evaluating Nanomedicine Pharmacokinetics?Drug Metab Dispos. 2016 Dec;44(12):1934-1939. doi: 10.1124/dmd.116.073148. Epub 2016 Sep 26. Drug Metab Dispos. 2016. PMID: 27670412 Free PMC article. Review.
References
-
- Cui Y, Andersen DK. Pancreatogenic diabetes: Special considerations for management. Pancreatology. 2011 Jul 9;11(3):279–94. - PubMed
-
- Yki-Jarvinen H, Kiviluoto T, Taskinen MR. Insulin resistance is a prominent feature of patients with pancreatogenic diabetes. Metabolism. 1986 Aug;35(8):718–27. - PubMed
-
- Nathan JD, Zdankiewicz PD, Wang J, Spector SA, Aspelund G, Jena BP, et al. Impaired hepatocyte glucose transport protein (GLUT2) internalization in chronic pancreatitis. Pancreas. 2001 Mar;22(2):172–8. - PubMed
-
- Cui Y, Andersen DK. Diabetes and Pancreatic Cancer. Endocr Relat Cancer. 2012;19(5):F9–26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

