Role of baseline HIV-1 DNA level in highly-experienced patients receiving raltegravir, etravirine and darunavir/ritonavir regimen (ANRS139 TRIO trial)
- PMID: 23349724
- PMCID: PMC3547918
- DOI: 10.1371/journal.pone.0053621
Role of baseline HIV-1 DNA level in highly-experienced patients receiving raltegravir, etravirine and darunavir/ritonavir regimen (ANRS139 TRIO trial)
Abstract
Objective: In the ANRS 139 TRIO trial, the use of 3 new active drugs (raltegravir, etravirine, and darunavir/ritonavir), resulted in a potent and sustained inhibition of viral replication in multidrug-resistant treatment-experienced patients. The aim of this virological sub-study of the ANRS 139 TRIO trial was to assess: (i) the evolution of HIV-1 DNA over the first year; and (ii) the association between baseline HIV-1 DNA and virological outcome.
Methods: Among the 103 HIV-1-infected patients included in the ANRS-139 TRIO trial, HIV-1 DNA specimens were available for 92, 84, 88, and 83 patients at Week (W)0, W12, W24, and W48, respectively. Quantification of total HIV-1 DNA was performed by using the commercial kit "Generic HIV DNA Cell" (Biocentric, Bandol, France).
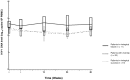
Results: Baseline median HIV-1 DNA of patients displaying virological success (n= 61), viral blip (n= 20), and virological failure (n = 11) were 2.34 log(10) copies/10(6) PBMC (IQR= 2.15-2.66), 2.42 (IQR = 2.12-2.48), and 2.68 (IQR= 2.46-2.83), respectively. Although not statistically significant, patients exhibiting virological success or viral blip had a tendency to display lower baseline HIV-1 DNA than patients experiencing virological failure (P = 0.06). Median decrease of HIV-1 DNA between baseline and W48 was -0.13 log(10) copies/10(6) PBMC (IQR = -0.34 to +0.10), mainly explained by the evolution from W0 to W4. No more changes were observed in the W4-W48 period.
Conclusions: In highly-experienced multidrug-resistant patients, HIV-1 DNA slightly decreased during the first month and then remained stable during the first year of highly potent antiretroviral regimen. In this population, baseline HIV-1 DNA might help to better predict the virological response and to tailor clinical therapeutic management as more aggressive therapeutic choices in patients with higher baseline HIV-1 DNA.
Conflict of interest statement
Figures
Similar articles
-
Resistance analyses in highly experienced patients failing raltegravir, etravirine and darunavir/ritonavir regimen.AIDS. 2010 Nov 13;24(17):2651-6. doi: 10.1097/QAD.0b013e32833ed2a7. AIDS. 2010. PMID: 20802293 Clinical Trial.
-
High rate of virologic suppression with raltegravir plus etravirine and darunavir/ritonavir among treatment-experienced patients infected with multidrug-resistant HIV: results of the ANRS 139 TRIO trial.Clin Infect Dis. 2009 Nov 1;49(9):1441-9. doi: 10.1086/630210. Clin Infect Dis. 2009. PMID: 19814627 Clinical Trial.
-
Highly frequent HIV-1 minority resistant variants at baseline of the ANRS 139 TRIO trial had a limited impact on virological response.J Antimicrob Chemother. 2015 Jul;70(7):2090-6. doi: 10.1093/jac/dkv048. Epub 2015 Mar 8. J Antimicrob Chemother. 2015. PMID: 25755001
-
Novel antiretroviral combinations in treatment-experienced patients with HIV infection: rationale and results.Drugs. 2010 Sep 10;70(13):1629-42. doi: 10.2165/11538020-000000000-00000. Drugs. 2010. PMID: 20731472 Review.
-
[Darunavir in patients with advanced HIV and multiresistance. The POWER, DUET and BENCHMRK studies].Enferm Infecc Microbiol Clin. 2008 Oct;26 Suppl 10:23-31. doi: 10.1016/s0213-005x(08)76550-9. Enferm Infecc Microbiol Clin. 2008. PMID: 19195456 Review. Spanish.
Cited by
-
HIV Replication at Low Copy Number and its Correlation with the HIV Reservoir: A Clinical Perspective.Curr HIV Res. 2015;13(3):250-7. doi: 10.2174/1570162x13666150407142539. Curr HIV Res. 2015. PMID: 25845389 Free PMC article. Review.
-
Integrase Strand Transfer Inhibitors in HIV Therapy.Infect Dis Ther. 2013 Dec;2(2):83-93. doi: 10.1007/s40121-013-0020-8. Epub 2013 Nov 19. Infect Dis Ther. 2013. PMID: 25134473 Free PMC article.
References
-
- Goujard C, Bonarek M, Meyer L, Bonnet F, Chaix ML, et al. (2006) CD4 cell count and HIV DNA level are independent predictors of disease progression after primary HIV type 1 infection in untreated patients. Clin Infect Dis 42: 709–715. - PubMed
-
- Rouzioux C, Hubert JB, Burgard M, Deveau C, Goujard C, et al. (2005) Early levels of HIV-1 DNA in peripheral blood mononuclear cells are predictive of disease progression independently of HIV-1 RNA levels and CD4+ T-cell counts. J Infect Dis 192: 46–55. - PubMed
-
- Lambert-Niclot S, Flandre P, Valantin MA, Peytavin G, Duvivier C, et al. (2011) Factors associated with virological failure in HIV-1-infected patients receiving darunavir/ritonavir monotherapy. J Infect Dis 204: 1211–1216. - PubMed
-
- Yazdanpanah Y, Fagard C, Descamps D, Taburet AM, Colin C, et al. (2009) High rate of virologic suppression with raltegravir plus etravirine and darunavir/ritonavir among treatment-experienced patients infected with multidrug-resistant HIV: results of the ANRS 139 TRIO trial. Clin Infect Dis 49: 1441–1449. - PubMed
-
- Fagard C, Colin C, Charpentier C, Rami A, Jacomet C, et al. (2012) Long-term efficacy and safety of raltegravir, etravirine, and darunavir/ritonavir in treatment-experienced patients: week 96 results from the ANRS 139 TRIO trial. J Acquir Immune Defic Syndr 59: 489–493. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical