Distinct profiles of effector cytokines mark the different phases of Crohn's disease
- PMID: 23349929
- PMCID: PMC3547873
- DOI: 10.1371/journal.pone.0054562
Distinct profiles of effector cytokines mark the different phases of Crohn's disease
Abstract
Objective: Crohn's Disease (CD)-associated inflammation is supposed to be driven by T helper (Th)1/Th17 cell-derived cytokines, even though there is evidence that the mucosal profile of cytokine may vary with the evolution of the disease. We aimed at comparing the pattern of effector cytokines in early and established lesions of CD.
Design: Mucosal samples were taken from the neo-terminal ileum of CD patients undergoing ileocolonic resection, with (early lesions) or without post-operative recurrence, and terminal ileum of CD patients with long-standing disease undergoing intestinal resection (established lesions). Inflammatory cell infiltrate was examined by immunofluorescence and cytokine expression was analysed by real-time PCR, flow-cytometry and ELISA.
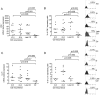
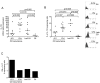
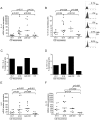
Results: Before the appearance of endoscopic lesions, the mucosa of the neo-terminal ileum contained high number of T cells and macrophages, elevated levels of Th1-related cytokines and TNF-α and slightly increased IL-17A expression. Transition from this stage to endoscopic recurrence was marked by abundance of Th1 cytokines, marked increase in IL-17A, and induction of IL-6 and IL-23, two cytokines involved in the control of Th17 cell responses. In samples with established lesions, there was a mixed Th1/Th17 response with no TNF-α induction. Expression of IL-4 and IL-5 was up-regulated in both early and established lesions even though the fraction of IL-4-producing cells was lower than that of cells producing either interferon-γ or IL-17A.
Conclusions: Distinct mucosal profiles of cytokines are produced during the different phases of CD. A better understanding of the cytokines temporally regulated in CD tissue could help optimize therapeutic interventions in CD.
Conflict of interest statement
Figures



References
-
- Fuss IJ, Neurath M, Boirivant M, Klein JS, de la Motte C, et al. (1996) Disparate CD4+ lamina propria (LP) lymphokine secretion profiles in inflammatory bowel disease. Crohn’s disease LP cells manifest increased secretion of IFN-gamma, whereas ulcerative colitis LP cells manifest increased secretion of IL-5. J Immunol 157: 1261–1270. - PubMed
-
- Monteleone G, Biancone L, Marasco R, Morrone G, Marasco O, et al. (1997) Interleukin 12 is expressed and actively released by Crohn’s disease intestinal lamina propria mononuclear cells. Gastroenterology 112: 1169–1178. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

