The non-peptidic part determines the internalization mechanism and intracellular trafficking of peptide amphiphiles
- PMID: 23349939
- PMCID: PMC3547919
- DOI: 10.1371/journal.pone.0054611
The non-peptidic part determines the internalization mechanism and intracellular trafficking of peptide amphiphiles
Abstract
Background: Peptide amphiphiles (PAs) are a class of amphiphilic molecules able to self-assemble into nanomaterials that have shown efficient in vivo targeted delivery. Understanding the interactions of PAs with cells and the mechanisms of their internalization and intracellular trafficking is critical in their further development for therapeutic delivery applications.
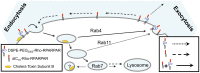
Methodology/principal findings: PAs of a novel, cell- and tissue-penetrating peptide were synthesized possessing two different lipophilic tail architectures and their interactions with prostate cancer cells were studied in vitro. Cell uptake of peptides was greatly enhanced post-modification. Internalization occurred via lipid-raft mediated endocytosis and was common for the two analogs studied. On the contrary, we identified the non-peptidic part as the determining factor of differences between intracellular trafficking and retention of PAs. PAs composed of di-stearyl lipid tails linked through poly(ethylene glycol) to the peptide exhibited higher exocytosis rates and employed different recycling pathways compared to ones consisting of di-palmitic-coupled peptides. As a result, cell association of the former PAs decreased with time.
Conclusions/significance: Control over peptide intracellular localization and retention is possible by appropriate modification with synthetic hydrophobic tails. We propose this as a strategy to design improved peptide-based delivery systems.
Conflict of interest statement
Figures




Similar articles
-
Cathepsin-Mediated Cleavage of Peptides from Peptide Amphiphiles Leads to Enhanced Intracellular Peptide Accumulation.Bioconjug Chem. 2017 Sep 20;28(9):2316-2326. doi: 10.1021/acs.bioconjchem.7b00364. Epub 2017 Aug 24. Bioconjug Chem. 2017. PMID: 28771332 Free PMC article.
-
Plasma membrane-anchorable photosensitizing nanomicelles for lipid raft-responsive and light-controllable intracellular drug delivery.J Control Release. 2018 Sep 28;286:103-113. doi: 10.1016/j.jconrel.2018.07.027. Epub 2018 Jul 17. J Control Release. 2018. PMID: 30026079
-
Multifunctional Self-Assembling Peptide-Based Nanostructures for Targeted Intracellular Delivery: Design, Physicochemical Characterization, and Biological Assessment.Methods Mol Biol. 2018;1758:11-26. doi: 10.1007/978-1-4939-7741-3_2. Methods Mol Biol. 2018. PMID: 29679319
-
Peptide Amphiphiles for Pharmaceutical Applications.Curr Med Chem. 2024;31(11):1332-1347. doi: 10.2174/0929867330666230408203820. Curr Med Chem. 2024. PMID: 37031390 Review.
-
Self-assembly of peptide amphiphiles for drug delivery: the role of peptide primary and secondary structures.Biomater Sci. 2017 Nov 21;5(12):2369-2380. doi: 10.1039/c7bm00730b. Biomater Sci. 2017. PMID: 29051950 Review.
Cited by
-
Peptide Amphiphiles as Biodegradable Adjuvants for Efficient Retroviral Gene Delivery.Adv Healthc Mater. 2024 Feb;13(4):e2301364. doi: 10.1002/adhm.202301364. Epub 2023 Nov 23. Adv Healthc Mater. 2024. PMID: 37947246 Free PMC article.
-
Inhibition of atherosclerosis-promoting microRNAs via targeted polyelectrolyte complex micelles.J Mater Chem B. 2014 Dec 14;2(46):8142-8153. doi: 10.1039/C4TB00977K. J Mater Chem B. 2014. PMID: 25685357 Free PMC article.
-
Self-assembling peptide-based building blocks in medical applications.Adv Drug Deliv Rev. 2017 Feb;110-111:65-79. doi: 10.1016/j.addr.2016.08.006. Epub 2016 Aug 14. Adv Drug Deliv Rev. 2017. PMID: 27535485 Free PMC article. Review.
-
Modular Peptide Amphiphile Micelles Improving an Antibody-Mediated Immune Response to Group A Streptococcus.ACS Biomater Sci Eng. 2017;3(2):144-152. doi: 10.1021/acsbiomaterials.6b00422. Epub 2016 Sep 28. ACS Biomater Sci Eng. 2017. PMID: 29242824 Free PMC article.
-
BH3-mimetics: recent developments in cancer therapy.J Exp Clin Cancer Res. 2021 Nov 9;40(1):355. doi: 10.1186/s13046-021-02157-5. J Exp Clin Cancer Res. 2021. PMID: 34753495 Free PMC article. Review.
References
-
- Ruoslahti E, Bhatia SN, Sailor MJ (2010) Targeting of drugs and nanoparticles to tumors. The Journal of Cell Biology 188: 759–768 doi:10.1083/jcb.200910104. - DOI - PMC - PubMed
-
- Teesalu T, Sugahara KN, Ruoslahti E (2012) Mapping of vascular ZIP codes by phage display. Meth Enzymol 503: 35–56 doi:10.1016/B978-0-12-396962-0.00002-1. - DOI - PubMed
-
- Foerg C, Merkle HP (2007) On the biomedical promise of cell penetrating peptides: Limits versus prospects. J Pharm Sci 97: 144–162 doi:10.1002/jps.21117. - DOI - PubMed
-
- Teesalu T, Sugahara KN, Kotamraju VR, Ruoslahti E (2009) C-end rule peptides mediate neuropilin-1-dependent cell, vascular, and tissue penetration. Proceedings of the National Academy of Sciences 106: 16157–16162 doi:10.1073/pnas.0908201106. - DOI - PMC - PubMed
-
- Roth L, Agemy L, Kotamraju VR, Braun G, Teesalu T, et al... (2011) Transtumoral targeting enabled by a novel neuropilin-binding peptide. Oncogene. doi:10.1038/onc.2011.537. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

