N-acetylglucosamine (GlcNAc) functions in cell signaling
- PMID: 23350039
- PMCID: PMC3551598
- DOI: 10.6064/2012/489208
N-acetylglucosamine (GlcNAc) functions in cell signaling
Abstract
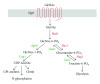
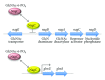
The amino sugar N-acetylglucosamine (GlcNAc) is well known for the important structural roles that it plays at the cell surface. It is a key component of bacterial cell wall peptidoglycan, fungal cell wall chitin, and the extracellular matrix of animal cells. Interestingly, recent studies have also identified new roles for GlcNAc in cell signaling. For example, GlcNAc stimulates the human fungal pathogen Candida albicans to undergo changes in morphogenesis and expression of virulence genes. Pathogenic E. coli respond to GlcNAc by altering the expression of fimbriae and CURLI fibers that promote biofilm formation and GlcNAc stimulates soil bacteria to undergo changes in morphogenesis and production of antibiotics. Studies with animal cells have revealed that GlcNAc influences cell signaling through the post-translational modification of proteins by glycosylation. O-linked attachment of GlcNAc to Ser and Thr residues regulates a variety of intracellular proteins, including transcription factors such as NFκB, c-myc and p53. In addition, the specificity of Notch family receptors for different ligands is altered by GlcNAc attachment to fucose residues in the extracellular domain. GlcNAc also impacts signal transduction by altering the degree of branching of N-linked glycans, which influences cell surface signaling proteins. These emerging roles of GlcNAc as an activator and mediator of cellular signaling in fungi, animals, and bacteria will be the focus of this review.
Figures

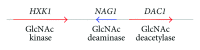
References
-
- Sohanpal BK, El-Labany S, Lahooti M, Plumbridge JA, Blomfield IC. Integrated regulatory responses of fimB to N-acetylneuraminic (sialic) acid and GlcNAc in Escherichia coli K-12. Proceedings of the National Academy of Sciences of the United States of America. 2004;101(46):16322–16327. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

