Olanzapine treatment of adolescent rats alters adult reward behaviour and nucleus accumbens function
- PMID: 23351612
- PMCID: PMC5819604
- DOI: 10.1017/S1461145712001642
Olanzapine treatment of adolescent rats alters adult reward behaviour and nucleus accumbens function
Abstract
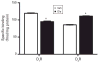
Antipsychotic drugs are increasingly used in children and adolescents to treat a variety of psychiatric disorders. However, little is known about the long-term effects of early life antipsychotic drug (APD) treatment. Most APDs are potent antagonists or partial agonists of dopamine (DA) D₂ receptors; atypical APDs also have multiple serotonergic activities. DA and serotonin regulate many neurodevelopmental processes. Thus, early life APD treatment can, potentially, perturb these processes, causing long-term behavioural and neurobiological sequelae. We treated adolescent, male rats with olanzapine (Ola) on post-natal days 28-49, under dosing conditions that approximate those employed therapeutically in humans. As adults, they exhibited enhanced conditioned place preference for amphetamine, as compared to vehicle-treated rats. In the nucleus accumbens core, DA D₁ receptor binding was reduced, D₂ binding was increased and DA release evoked by electrical stimulation of the ventral tegmental area was reduced. Thus, adolescent Ola treatment enduringly alters a key behavioural response to rewarding stimuli and modifies DAergic neurotransmission in the nucleus accumbens. The persistence of these changes suggests that even limited periods of early life Ola treatment may induce enduring changes in other reward-related behaviours and in behavioural and neurobiological responses to therapeutic and illicit psychotropic drugs. These results underscore the importance of improved understanding of the enduring sequelae of paediatric APD treatment as a basis for weighing the benefits and risks of adolescent APD therapy, especially prophylactic treatment in high-risk, asymptomatic patients.
Conflict of interest statement
None.
Figures

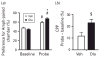

Similar articles
-
Olanzapine antipsychotic treatment of adolescent rats causes long term changes in glutamate and GABA levels in the nucleus accumbens.Schizophr Res. 2015 Feb;161(2-3):452-7. doi: 10.1016/j.schres.2014.10.034. Epub 2014 Dec 5. Schizophr Res. 2015. PMID: 25487700 Free PMC article.
-
Olanzapine treatment of adolescent rats causes enduring specific memory impairments and alters cortical development and function.PLoS One. 2013;8(2):e57308. doi: 10.1371/journal.pone.0057308. Epub 2013 Feb 20. PLoS One. 2013. PMID: 23437365 Free PMC article.
-
Antipsychotic treatment leading to dopamine supersensitivity persistently alters nucleus accumbens function.Neuropharmacology. 2015 Dec;99:715-25. doi: 10.1016/j.neuropharm.2015.03.012. Epub 2015 Mar 27. Neuropharmacology. 2015. PMID: 25823912
-
Chronic betahistine co-treatment reverses olanzapine's effects on dopamine D₂ but not 5-HT2A/2C bindings in rat brains.Prog Neuropsychopharmacol Biol Psychiatry. 2015 Jan 2;56:75-80. doi: 10.1016/j.pnpbp.2014.08.006. Epub 2014 Aug 20. Prog Neuropsychopharmacol Biol Psychiatry. 2015. PMID: 25149912
-
Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine.Behav Brain Res. 2002 Dec 2;137(1-2):3-25. doi: 10.1016/s0166-4328(02)00282-6. Behav Brain Res. 2002. PMID: 12445713 Review.
Cited by
-
The effects of amphetamine on working memory and locomotor activity in adult rats administered risperidone early in life.Behav Brain Res. 2019 Apr 19;362:64-70. doi: 10.1016/j.bbr.2018.12.044. Epub 2018 Dec 27. Behav Brain Res. 2019. PMID: 30594546 Free PMC article.
-
Long-lasting sensitization induced by repeated risperidone treatment in adolescent Sprague-Dawley rats: a possible D2 receptor mediated phenomenon?Psychopharmacology (Berl). 2014 Apr;231(8):1649-1659. doi: 10.1007/s00213-013-3386-0. Epub 2013 Dec 21. Psychopharmacology (Berl). 2014. PMID: 24363078 Free PMC article.
-
Olanzapine antipsychotic treatment of adolescent rats causes long term changes in glutamate and GABA levels in the nucleus accumbens.Schizophr Res. 2015 Feb;161(2-3):452-7. doi: 10.1016/j.schres.2014.10.034. Epub 2014 Dec 5. Schizophr Res. 2015. PMID: 25487700 Free PMC article.
-
Early antipsychotic treatment in juvenile rats elicits long-term alterations to the adult serotonin receptors.Neuropsychiatr Dis Treat. 2018 Jun 14;14:1569-1583. doi: 10.2147/NDT.S158545. eCollection 2018. Neuropsychiatr Dis Treat. 2018. PMID: 29950841 Free PMC article.
-
Sex differences in aripiprazole sensitization from adolescence to adulthood.Pharmacol Biochem Behav. 2017 May;156:39-47. doi: 10.1016/j.pbb.2017.04.001. Epub 2017 Apr 4. Pharmacol Biochem Behav. 2017. PMID: 28389140 Free PMC article.
References
-
- Andersen SL, Rutstein M, Benzo JM, Hostetter JC, Teicher MH. Sex differences in dopamine receptor overproduction and elimination. Neuroreport. 1997;8:1495–1498. - PubMed
-
- Andersen SL, Thompson AT, Rutstein M, Hostetter JC, Teicher MH. Dopamine receptor pruning in prefrontal cortex during the periadolescent period in rats. Synapse. 2000;37:167–169. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

