Models of drug-induced liver injury for evaluation of phytotherapeutics and other natural products
- PMID: 23353004
- PMCID: PMC3608827
- DOI: 10.1016/j.fct.2012.12.063
Models of drug-induced liver injury for evaluation of phytotherapeutics and other natural products
Abstract
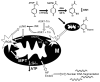
Extracts from medicinal plants, many of which have been used for centuries, are increasingly tested in models of hepatotoxicity. One of the most popular models to evaluate the hepatoprotective potential of natural products is acetaminophen (APAP)-induced liver injury, although other hepatotoxicity models such as carbon tetrachloride, thioacetamide, ethanol and endotoxin are occasionally used. APAP overdose is a clinically relevant model of drug-induced liver injury. Critical mechanisms and signaling pathways, which trigger necrotic cell death and sterile inflammation, are discussed. Although there is increasing understanding of the pathophysiology of APAP-induced liver injury, the mechanism is complex and prone to misinterpretation, especially when unknown chemicals such as plant extracts are tested. This review discusses the fundamental aspects that need to be considered when using this model, such as selection of the animal species or in vitro system, timing and dose-responses of signaling events, metabolic activation and protein adduct formation, the role of lipid peroxidation and apoptotic versus necrotic cell death, and the impact of the ensuing sterile inflammatory response. The goal is to enable researchers to select the appropriate model and experimental conditions for testing of natural products that will yield clinically relevant results and allow valid interpretations of the pharmacological mechanisms.
Copyright © 2013 Elsevier Ltd. All rights reserved.
Figures
References
-
- Adams ML, Pierce RH, Vail ME, White CC, Tonge RP, Kavanagh TJ, Fausto N, Nelson SD, Bruschi SA. Enhanced acetaminophen hepatotoxicity in transgenic mice overexpressing BCL-2. Mol. Pharmacol. 2001;60:907–915. - PubMed
-
- Ajith TA, Hema U, Aswathy MS. Zingiber officinale Roscoe prevents acetaminophen-induced acute hepatotoxicity by enhancing hepatic antioxidant status. Food Chem. Toxicol. 2007;45:2267–2272. - PubMed
-
- Antoine DJ, Jenkins RE, Dear JW, Williams DP, McGill MR, Sharpe MR, Craig DG, Simpson KJ, Jaeschke H, Park BK. Molecular forms of HMGB1 and keratin-18 as mechanistic biomarkers for mode of cell death and prognosis during clinical acetaminophen hepatotoxicity. J. Hepatol. 2012;56:1070–1079. - PMC - PubMed
-
- Antoine DJ, Williams DP, Kipar A, Jenkins RE, Regan SL, Sathish JG, Kitteringham NR, Park BK. High-mobility group box-1 protein and keratin-18, circulating serum proteins informative of acetaminophen-induced necrosis and apoptosis in vivo. Toxicol. Sci. 2009;112:521–531. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

