Oxygen tension modulates differentiation and primary macrophage functions in the human monocytic THP-1 cell line
- PMID: 23355903
- PMCID: PMC3552948
- DOI: 10.1371/journal.pone.0054926
Oxygen tension modulates differentiation and primary macrophage functions in the human monocytic THP-1 cell line
Abstract
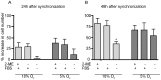
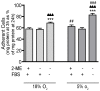
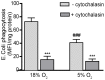
The human THP-1 cell line is widely used as an in vitro model system for studying macrophage differentiation and function. Conventional culture conditions for these cells consist of ambient oxygen pressure (∼20% v/v) and medium supplemented with the thiol 2-mercaptoethanol (2-ME) and serum. In consideration of the redox activities of O₂ and 2-ME, and the extensive experimental evidence supporting a role for reactive oxygen species (ROS) in the differentiation and function of macrophages, we addressed the question of whether culturing THP-1 cells under a more physiologically relevant oxygen tension (5% O₂) in the absence of 2-ME and serum would alter THP-1 cell physiology. Comparisons of cultures maintained in 18% O₂versus 5% O₂ indicated that reducing oxygen tension had no effect on the proliferation of undifferentiated THP-1 cells. However, decreasing the oxygen tension to 5% O₂ significantly increased the rate of phorbol ester-induced differentiation of THP-1 cells into macrophage-like cells as well as the metabolic activity of both undifferentiated and PMA-differentiated THP-1 cells. Removal of both 2-ME and serum from the medium decreased the proliferation of undifferentiated THP-1 cells but increased metabolic activity and the rate of differentiation under either oxygen tension. In differentiated THP-1 cells, lowering the oxygen tension to 5% O₂ decreased phagocytic activity, the constitutive release of β-hexosaminidase and LPS-induced NF-κB activation but enhanced LPS-stimulated release of cytokines. Collectively, these data demonstrate that oxygen tension influences THP-1 cell differentiation and primary macrophage functions, and suggest that culturing these cells under tightly regulated oxygen tension in the absence of exogenous reducing agent and serum is likely to provide a physiologically relevant baseline from which to study the role of the local redox environment in regulating THP-1 cell physiology.
Conflict of interest statement
Figures




Similar articles
-
LPS induces hypoxia-inducible factor 1 activation in macrophage-differentiated cells in a reactive oxygen species-dependent manner.Antioxid Redox Signal. 2008 May;10(5):983-95. doi: 10.1089/ars.2007.1825. Antioxid Redox Signal. 2008. PMID: 18199003
-
The Trace Element Selenium Is Important for Redox Signaling in Phorbol Ester-Differentiated THP-1 Macrophages.Int J Mol Sci. 2021 Oct 14;22(20):11060. doi: 10.3390/ijms222011060. Int J Mol Sci. 2021. PMID: 34681720 Free PMC article.
-
Rhizoctonia bataticola lectin (RBL) induces phenotypic and functional characteristics of macrophages in THP-1 cells and human monocytes.Immunol Lett. 2015 Feb;163(2):163-72. doi: 10.1016/j.imlet.2014.12.005. Epub 2014 Dec 30. Immunol Lett. 2015. PMID: 25555439
-
High Uric Acid Activates the ROS-AMPK Pathway, Impairs CD68 Expression and Inhibits OxLDL-Induced Foam-Cell Formation in a Human Monocytic Cell Line, THP-1.Cell Physiol Biochem. 2016;40(3-4):538-548. doi: 10.1159/000452567. Epub 2016 Nov 25. Cell Physiol Biochem. 2016. PMID: 27889764
-
Decades Long Involvement of THP-1 Cells as a Model for Macrophage Research: A Comprehensive Review.Antiinflamm Antiallergy Agents Med Chem. 2024;23(2):85-104. doi: 10.2174/0118715230294413240415054610. Antiinflamm Antiallergy Agents Med Chem. 2024. PMID: 38676532 Review.
Cited by
-
The Role of Tissue Oxygen Tension in Dengue Virus Replication.Cells. 2018 Dec 1;7(12):241. doi: 10.3390/cells7120241. Cells. 2018. PMID: 30513781 Free PMC article.
-
Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition)1.Autophagy. 2021 Jan;17(1):1-382. doi: 10.1080/15548627.2020.1797280. Epub 2021 Feb 8. Autophagy. 2021. PMID: 33634751 Free PMC article.
-
Immunostimulant Bathing Influences the Expression of Immune- and Metabolic-Related Genes in Atlantic Salmon Alevins.Biology (Basel). 2021 Sep 29;10(10):980. doi: 10.3390/biology10100980. Biology (Basel). 2021. PMID: 34681079 Free PMC article.
-
An improved organotypic cell culture system to study tissue-resident macrophages ex vivo.Cell Rep Methods. 2022 Jul 26;2(8):100260. doi: 10.1016/j.crmeth.2022.100260. eCollection 2022 Aug 22. Cell Rep Methods. 2022. PMID: 36046625 Free PMC article.
-
Hydrogen peroxide-induced oxidative stress and its impact on innate immune responses in lung carcinoma A549 cells.Mol Cell Biochem. 2019 Jan;450(1-2):135-147. doi: 10.1007/s11010-018-3380-2. Epub 2018 Jun 25. Mol Cell Biochem. 2019. PMID: 29938378
References
-
- Tsuchiya S, Yamabe M, Yamaguchi Y, Kobayashi Y, Konno T, et al. (1980) Establishment and characterization of a human acute monocytic leukemia cell line (THP-1). Int J Cancer 26: 171–176. - PubMed
-
- Csete M (2005) Oxygen in the cultivation of stem cells. Ann N Y Acad Sci 1049: 1–8. - PubMed
-
- Meister A, Anderson ME (1983) Glutathione. Annu Rev Biochem 52: 711–760. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

