MicroRNA Profiling Reveals Distinct Mechanisms Governing Cardiac and Neural Lineage-Specification of Pluripotent Human Embryonic Stem Cells
- PMID: 23355957
- PMCID: PMC3554249
- DOI: 10.4172/2157-7633.1000124
MicroRNA Profiling Reveals Distinct Mechanisms Governing Cardiac and Neural Lineage-Specification of Pluripotent Human Embryonic Stem Cells
Abstract
Realizing the potential of human embryonic stem cells (hESCs) has been hindered by the inefficiency and instability of generating desired cell types from pluripotent cells through multi-lineage differentiation. We recently reported that pluripotent hESCs maintained under a defined platform can be uniformly converted into a cardiac or neural lineage by small molecule induction, which enables lineage-specific differentiation direct from the pluripotent state of hESCs and opens the door to investigate human embryonic development using in vitro cellular model systems. To identify mechanisms of small molecule induced lineage-specification of pluripotent hESCs, in this study, we compared the expression and intracellular distribution patterns of a set of cardinal chromatin modifiers in pluripotent hESCs, nicotinamide (NAM)-induced cardiomesodermal cells, and retinoic acid (RA)-induced neuroectodermal cells. Further, genome-scale profiling of microRNA (miRNA) differential expression patterns was used to monitor the regulatory networks of the entire genome and identify the development-initiating miRNAs in hESC cardiac and neural lineage-specification. We found that NAM induced nuclear translocation of NAD-dependent histone deacetylase SIRT1 and global chromatin silencing, while RA induced silencing of pluripotence-associated hsa-miR-302 family and drastic up-regulation of neuroectodermal Hox miRNA hsa-miR-10 family to high levels. Genome-scale miRNA profiling indentified that a unique set of pluripotence-associated miRNAs was down-regulated, while novel sets of distinct cardiac- and neural-driving miRNAs were up-regulated upon the induction of lineage-specification direct from the pluripotent state of hESCs. These findings suggest that a predominant epigenetic mechanism via SIRT1-mediated global chromatin silencing governs NAM-induced hESC cardiac fate determination, while a predominant genetic mechanism via silencing of pluripotence-associated hsa-miR-302 family and drastic up-regulation of neuroectodermal Hox miRNA hsa-miR-10 family governs RA-induced hESC neural fate determination. This study provides critical insight into the earliest events in human embryogenesis as well as offers means for small molecule-mediated direct control and modulation of hESC pluripotent fate when deriving clinically-relevant lineages for regenerative therapies.
Figures
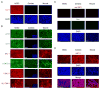
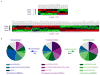
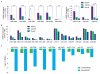
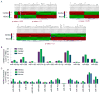
Similar articles
-
Embedding the Future of Regenerative Medicine into the Open Epigenomic Landscape of Pluripotent Human Embryonic Stem Cells.Annu Res Rev Biol. 2013 Oct;3(4):323-349. Annu Res Rev Biol. 2013. PMID: 25309947 Free PMC article.
-
Genome-Scale Mapping of MicroRNA Signatures in Human Embryonic Stem Cell Neurogenesis.Mol Med Ther. 2012 Dec 10;1(2):105. doi: 10.4172/2324-8769.1000105. Mol Med Ther. 2012. PMID: 23543894 Free PMC article.
-
Constraining the Pluripotent Fate of Human Embryonic Stem Cells for Tissue Engineering and Cell Therapy - The Turning Point of Cell-Based Regenerative Medicine.Br Biotechnol J. 2013 Oct 1;3(4):424-457. doi: 10.9734/BBJ/2013/4309#sthash.6D8Rulbv.dpuf. Br Biotechnol J. 2013. PMID: 24926434 Free PMC article.
-
Embryonic stem cell microRNAs: defining factors in induced pluripotent (iPS) and cancer (CSC) stem cells?Curr Stem Cell Res Ther. 2009 Sep;4(3):168-77. doi: 10.2174/157488809789057400. Curr Stem Cell Res Ther. 2009. PMID: 19492978 Review.
-
Lnc-ing pluripotency maintenance and early differentiation in human pluripotent stem cells.FASEB J. 2021 Apr;35(4):e21438. doi: 10.1096/fj.202002278R. FASEB J. 2021. PMID: 33749897 Review.
Cited by
-
Embedding the Future of Regenerative Medicine into the Open Epigenomic Landscape of Pluripotent Human Embryonic Stem Cells.Annu Res Rev Biol. 2013 Oct;3(4):323-349. Annu Res Rev Biol. 2013. PMID: 25309947 Free PMC article.
-
SIRT1 and Neural Cell Fate Determination.Mol Neurobiol. 2016 Jul;53(5):2815-2825. doi: 10.1007/s12035-015-9158-6. Epub 2015 Apr 8. Mol Neurobiol. 2016. PMID: 25850787 Review.
-
MicroRNAs: modulators of cell identity, and their applications in tissue engineering.Microrna. 2014;3(1):45-53. doi: 10.2174/2211536603666140522003539. Microrna. 2014. PMID: 25069512 Free PMC article. Review.
-
Effects of erythropoietin in murine-induced pluripotent cell-derived panneural progenitor cells.Mol Med. 2013 Nov 8;19(1):399-408. doi: 10.2119/molmed.2013.00136. Mol Med. 2013. PMID: 24408113 Free PMC article.
-
Assessment of the Potential of CDK2 Inhibitor NU6140 to Influence the Expression of Pluripotency Markers NANOG, OCT4, and SOX2 in 2102Ep and H9 Cells.Int J Cell Biol. 2014;2014:280638. doi: 10.1155/2014/280638. Epub 2014 Nov 17. Int J Cell Biol. 2014. PMID: 25477962 Free PMC article.
References
-
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. - PubMed
-
- Lee G, Kim H, Elkabetz Y, Al Shamy G, Panagiotakos G, et al. Isolation and directed differentiation of neural crest stem cells derived from human embryonic stem cells. Nat Biotechnol. 2007;25:1468–1475. - PubMed
-
- Roy NS, Cleren C, Singh SK, Yang L, Beal MF, et al. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nat Med. 2006;12:1259–1268. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
