M98K-OPTN induces transferrin receptor degradation and RAB12-mediated autophagic death in retinal ganglion cells
- PMID: 23357852
- PMCID: PMC3627667
- DOI: 10.4161/auto.23458
M98K-OPTN induces transferrin receptor degradation and RAB12-mediated autophagic death in retinal ganglion cells
Abstract
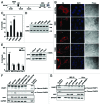
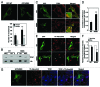
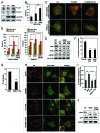
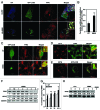
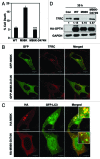
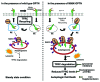
Mutations in the autophagy receptor OPTN/optineurin are associated with the pathogenesis of glaucoma and amyotrophic lateral sclerosis, but the underlying molecular basis is poorly understood. The OPTN variant, M98K has been described as a risk factor for normal tension glaucoma in some ethnic groups. Here, we examined the consequence of the M98K mutation in affecting cellular functions of OPTN. Overexpression of M98K-OPTN induced death of retinal ganglion cells (RGC-5 cell line), but not of other neuronal and non-neuronal cells. Enhanced levels of the autophagy marker, LC3-II, a post-translationally modified form of LC3, in M98K-OPTN-expressing cells and the inability of an LC3-binding-defective M98K variant of OPTN to induce cell death, suggested that autophagy contributes to cell death. Knockdown of Atg5 reduced M98K-induced death of RGC-5 cells, further supporting the involvement of autophagy. Overexpression of M98K-OPTN enhanced autophagosome formation and potentiated the delivery of transferrin receptor to autophagosomes for degradation resulting in reduced cellular transferrin receptor levels. Coexpression of transferrin receptor or supplementation of media with an iron donor reduced M98K-induced cell death. OPTN complexes with RAB12, a GTPase involved in vesicle trafficking, and M98K variant shows enhanced colocalization with RAB12. Knockdown of Rab12 increased transferrin receptor level and reduced M98K-induced cell death. RAB12 is present in autophagosomes and knockdown of Rab12 resulted in reduced formation of autolysosomes during starvation-induced autophagy, implicating a role for RAB12 in autophagy. These results also show that transferrin receptor degradation and autophagy play a crucial role in RGC-5 cell death induced by M98K variant of OPTN.
Keywords: RAB12; autophagy; glaucoma; optineurin; retinal ganglion cells; transferrin receptor.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
