Exemestane versus anastrozole in postmenopausal women with early breast cancer: NCIC CTG MA.27--a randomized controlled phase III trial
- PMID: 23358971
- PMCID: PMC3612593
- DOI: 10.1200/JCO.2012.44.7805
Exemestane versus anastrozole in postmenopausal women with early breast cancer: NCIC CTG MA.27--a randomized controlled phase III trial
Abstract
Purpose: In patients with hormone-dependent postmenopausal breast cancer, standard adjuvant therapy involves 5 years of the nonsteroidal aromatase inhibitors anastrozole and letrozole. The steroidal inhibitor exemestane is partially non-cross-resistant with nonsteroidal aromatase inhibitors and is a mild androgen and could prove superior to anastrozole regarding efficacy and toxicity, specifically with less bone loss.
Patients and methods: We designed an open-label, randomized, phase III trial of 5 years of exemestane versus anastrozole with a two-sided test of superiority to detect a 2.4% improvement with exemestane in 5-year event-free survival (EFS). Secondary objectives included assessment of overall survival, distant disease-free survival, incidence of contralateral new primary breast cancer, and safety.
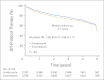
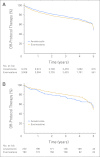
Results: In the study, 7,576 women (median age, 64.1 years) were enrolled. At median follow-up of 4.1 years, 4-year EFS was 91% for exemestane and 91.2% for anastrozole (stratified hazard ratio, 1.02; 95% CI, 0.87 to 1.18; P = .85). Overall, distant disease-free survival and disease-specific survival were also similar. In all, 31.6% of patients discontinued treatment as a result of adverse effects, concomitant disease, or study refusal. Osteoporosis/osteopenia, hypertriglyceridemia, vaginal bleeding, and hypercholesterolemia were less frequent on exemestane, whereas mild liver function abnormalities and rare episodes of atrial fibrillation were less frequent on anastrozole. Vasomotor and musculoskeletal symptoms were similar between arms.
Conclusion: This first comparison of steroidal and nonsteroidal classes of aromatase inhibitors showed neither to be superior in terms of breast cancer outcomes as 5-year initial adjuvant therapy for postmenopausal breast cancer by two-way test. Less toxicity on bone is compatible with one hypothesis behind MA.27 but requires confirmation. Exemestane should be considered another option as up-front adjuvant therapy for postmenopausal hormone receptor-positive breast cancer.
Trial registration: ClinicalTrials.gov NCT00066573.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures

Comment in
-
Aromatase inhibitors and safety: clinical or statistical significance?J Clin Oncol. 2013 Sep 20;31(27):3439. doi: 10.1200/JCO.2013.49.2884. Epub 2013 Aug 5. J Clin Oncol. 2013. PMID: 23918949 No abstract available.
-
Reply to e. Esin et al.J Clin Oncol. 2013 Sep 20;31(27):3439-40. doi: 10.1200/JCO.2013.51.4893. Epub 2013 Aug 5. J Clin Oncol. 2013. PMID: 23943834 No abstract available.
References
-
- Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: Results of the ATAC (Arimidex, Tamoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer. 2003;98:1802–1810. - PubMed
-
- Breast International Group (BIG) 1-98 Collaborative Group. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353:2747–2757. - PubMed
-
- Dowsett M, Jack Cuzick J, Ingle J, et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28:509–518. - PubMed
-
- Coombes RC, Hall E, Gibson LJ, et al. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med. 2004;350:1081–1092. - PubMed
-
- van de Velde CJ, Rea D, Seynaeve C, et al. Adjuvant tamoxifen and exemestane in early breast cancer (TEAM): A randomized phase 3 trial. Lancet. 2011;377:321–331. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

