Interferon-γ promotes vascular remodeling in human microvascular endothelial cells by upregulating endothelin (ET)-1 and transforming growth factor (TGF) β2
- PMID: 23359533
- PMCID: PMC4072032
- DOI: 10.1002/jcp.24337
Interferon-γ promotes vascular remodeling in human microvascular endothelial cells by upregulating endothelin (ET)-1 and transforming growth factor (TGF) β2
Abstract
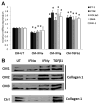
Systemic sclerosis (SSc) is a complex disease characterized by vascular alterations, activation of the immune system and tissue fibrosis. Previous studies have implicated activation of the interferon pathways in the pathogenesis of SSc. The goal of this study was to determine whether interferon type I and/or type II could play a pathogenic role in SSc vasculopathy. Human dermal microvascular endothelial cells (HDMVECs) and fibroblasts were obtained from foreskins of healthy newborns. The RT Profiler PCR Array System was utilized to screen for EndoMT genes. Treatment with IFN-α or IFN-γ downregulated Fli1 and VE-cadherin. In contrast, IFN-α and IFN-γ exerted opposite effects on the expression of α-SMA, CTGF, ET-1, and TGFβ2, with IFN-α downregulating and IFN-γ upregulating this set of genes. Blockade of TGFβ signaling normalized IFN-γ-mediated changes in Fli1, VE-cadherin, CTGF, and ET-1 levels, whereas upregulation of α-SMA and TGFβ2 was not affected. Bosentan treatment was more effective than TGFβ blockade in reversing the actions of IFN-γ, including downregulation of α-SMA and TGFβ2, suggesting that activation of the ET-1 pathway plays a main role in the IFN-γ responses in HDMECs. IFN-γ induced expression of selected genes related to endothelial-to-mesenchymal transition (EndoMT), including Snail1, FN1, PAI1, TWIST1, STAT3, RGS2, and components of the WNT pathway. The effect of IFN-γ on EndoMT was mediated via TGFβ2 and ET-1 signaling pathways. This study demonstrates distinct effects of IFN-α and IFN-γ on the biology of vascular endothelial cells. IFN-γ may contribute to abnormal vascular remodeling and fibrogenesis in SSc, partially via induction of EndoMT.
Copyright © 2013 Wiley Periodicals, Inc.
Conflict of interest statement
The authors declared that they have no conflicts of interest.
Figures





References
-
- Antonelli A, Ferri C, Fallahi P, Ferrari SM, Giuggioli D, Colaci M, Manfredi A, Frascerra S, Franzoni F, Galetta F, Ferrannini E. CXCL10 (alpha) and CCL2 (beta) chemokines in systemic sclerosis—A longitudinal study. Rheumatology (Oxford) 2008;47:45–49. - PubMed
-
- Antonelli A, Ferri C, Ferrari SM, Mancusi C, Colaci M, Sebastiani M, Fallahi P. IFN-gamma and TNF-alpha induce a different modulation of interleukin-6 in systemic sclerosis fibroblasts compared to healthy controls. Scand J Rheumatol. 2011;40:453–456. - PubMed
-
- Bellisai F, Morozzi G, Scaccia F, Chellini F, Simpatico A, Pecetti G, Galeazzi M. Evaluation of the effect of Bosentan treatment on proinflammatory cytokine serum levels in patients affected by Systemic Sclerosis. Int J Immunopathol Pharmacol. 2011;24:261–264. - PubMed
-
- Billiau A, Matthys P. Interferon-gamma: A historical perspective. Cytokine Growth Factor Rev. 2009;20:97–113. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

