Extracellular matrix-associated gene expression in adult sensory neuron populations cultured on a laminin substrate
- PMID: 23360524
- PMCID: PMC3610289
- DOI: 10.1186/1471-2202-14-15
Extracellular matrix-associated gene expression in adult sensory neuron populations cultured on a laminin substrate
Abstract
Background: In our previous investigations of the role of the extracellular matrix (ECM) in promoting neurite growth we have observed that a permissive laminin (LN) substrate stimulates differential growth responses in subpopulations of mature dorsal root ganglion (DRG) neurons. DRG neurons expressing Trk and p75 receptors grow neurites on a LN substrate in the absence of neurotrophins, while isolectin B4-binding neurons (IB4+) do not display significant growth under the same conditions. We set out to determine whether there was an expression signature of the LN-induced neurite growth phenotype. Using a lectin binding protocol IB4+ neurons were isolated from dissociated DRG neurons, creating two groups - IB4+ and IB4-. A small-scale microarray approach was employed to screen the expression of a panel of ECM-associated genes following dissociation (t=0) and after 24 hr culture on LN (t=24LN). This was followed by qRT-PCR and immunocytochemistry of selected genes.
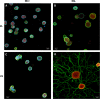
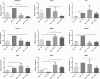
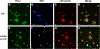
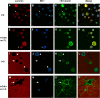
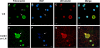
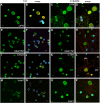
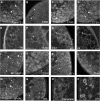
Results: The microarray screen showed that 36 of the 144 genes on the arrays were consistently expressed by the neurons. The array analyses showed that six genes had lower expression in the IB4+ neurons compared to the IB4- cells at t=0 (CTSH, Icam1, Itgβ1, Lamb1, Plat, Spp1), and one gene was expressed at higher levels in the IB4+ cells (Plaur). qRT-PCR was carried out as an independent assessment of the array results. There were discrepancies between the two methods, with qRT-PCR confirming the differences in Lamb1, Plat and Plaur, and showing decreased expression of AdamTs1, FN, and Icam in the IB4+ cells at t=0. After 24 hr culture on LN, there were no significant differences detected by qRT-PCR between the IB4+ and IB4- cells. However, both groups showed upregulation of Itgβ1 and Plaur after 24 hr on LN, the IB4+ group also had increased Plat, and the IB4- cells showed decreased Lamb1, Icam1 and AdamTs1. Further, the array screen also detected a number of genes (not subjected to qRT-PCR) expressed similarly by both populations in relatively high levels but not detectably influenced by time in culture (Bsg, Cst3, Ctsb, Ctsd, Ctsl, Mmp14, Mmp19, Sparc. We carried out immunohistochemistry to confirm expression of proteins encoded by a number of these genes.
Conclusions: Our results show that 1B4+ and IB4- neurons differ in the expression of several genes that are associated with responsiveness to the ECM prior to culturing (AdamTs1, FN, Icam1, Lamb1, Plat, Plaur). The data suggest that the genes expressed at higher levels in the IB4- neurons could contribute to the initial growth response of these cells in a permissive environment and could also represent a common injury response that subsequently promotes axon regeneration. The differential expression of several extracellular matrix molecules (FN, Lamb1, Icam) may suggest that the IB4- neurons are capable of maintaining /secreting their local extracellular environment which could aid in the regenerative process. Overall, these data provide new information on potential targets that could be manipulated to enhance axonal regeneration in the mature nervous system.
Figures


Similar articles
-
Impaired axonal regeneration by isolectin B4-binding dorsal root ganglion neurons in vitro.J Neurosci. 2007 Jan 31;27(5):1190-9. doi: 10.1523/JNEUROSCI.5089-06.2007. J Neurosci. 2007. PMID: 17267575 Free PMC article.
-
Laminin and growth factor receptor activation stimulates differential growth responses in subpopulations of adult DRG neurons.Eur J Neurosci. 2006 Aug;24(3):676-90. doi: 10.1111/j.1460-9568.2006.04963.x. Eur J Neurosci. 2006. PMID: 16930399
-
Growth cone behavior in the presence of soluble chondroitin sulfate proteoglycan (CSPG), compared to behavior on CSPG bound to laminin or fibronectin.Int J Dev Neurosci. 1996 Jun;14(3):331-49. doi: 10.1016/0736-5748(96)00017-2. Int J Dev Neurosci. 1996. PMID: 8842808
-
In vitro studies on the control of nerve fiber growth by the extracellular matrix of the nervous system.J Physiol (Paris). 1987;82(4):258-70. J Physiol (Paris). 1987. PMID: 3332689 Review.
-
Distribution and immunocytochemical characterization of dorsal root ganglion neurons innervating the lumbar intervertebral disc in rats: a review.Life Sci. 2004 Apr 9;74(21):2627-42. doi: 10.1016/j.lfs.2004.01.008. Life Sci. 2004. PMID: 15041445 Review.
Cited by
-
Networks of blood proteins in the neuroimmunology of schizophrenia.Transl Psychiatry. 2018 Jun 6;8(1):112. doi: 10.1038/s41398-018-0158-y. Transl Psychiatry. 2018. PMID: 29875399 Free PMC article.
-
Gene expression profiling of early hepatic stellate cell activation reveals a role for Igfbp3 in cell migration.PLoS One. 2013 Dec 17;8(12):e84071. doi: 10.1371/journal.pone.0084071. eCollection 2013. PLoS One. 2013. PMID: 24358328 Free PMC article.
-
The longitudinal transcriptomic response of the substantia nigra to intrastriatal 6-hydroxydopamine reveals significant upregulation of regeneration-associated genes.PLoS One. 2015 May 20;10(5):e0127768. doi: 10.1371/journal.pone.0127768. eCollection 2015. PLoS One. 2015. PMID: 25992874 Free PMC article.
-
Dorsal root ganglia neurons and differentiated adipose-derived stem cells: an in vitro co-culture model to study peripheral nerve regeneration.J Vis Exp. 2015 Feb 26;(96):52543. doi: 10.3791/52543. J Vis Exp. 2015. PMID: 25742570 Free PMC article.
-
Prognostic biomarkers and therapeutic targets in oral squamous cell carcinoma: a study based on cross-database analysis.Hereditas. 2021 Apr 23;158(1):15. doi: 10.1186/s41065-021-00181-1. Hereditas. 2021. PMID: 33892811 Free PMC article.
References
-
- Petruska JC, Napaporn J, Johnson RD, Gu JG, Cooper BY. Subclassified acutely dissociated cells of rat DRG: histochemistry and patterns of capsaicin-, proton-, and ATP-activated currents. J Neurophysiol. 2000;84(5):2365–2379. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

