Pitx2-mediated cardiac outflow tract remodeling
- PMID: 23361844
- PMCID: PMC3673775
- DOI: 10.1002/dvdy.23934
Pitx2-mediated cardiac outflow tract remodeling
Abstract
Background: Heart morphogenesis involves sequential anatomical changes from a linear tube of a single channel peristaltic pump to a four-chamber structure with two channels controlled by one-way valves. The developing heart undergoes continuous remodeling, including septation.
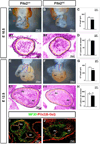
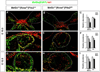
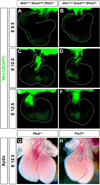
Results: Pitx2-null mice are characterized by cardiac septational defects of the atria, ventricles, and outflow tract. Pitx2-null mice also exhibited a short outflow tract, including unseptated conus and deformed endocardial cushions. Cushions were characterized with a jelly-like structure, rather than the distinct membrane-looking leaflets, indicating that endothelial mesenchymal transition was impaired in Pitx2(-/-) embryos. Mesoderm cells from the branchial arches and neural crest cells from the otic region contribute to the development of the endocardial cushions, and both were reduced in number. Members of the Fgf and Bmp families exhibited altered expression levels in the mutants.
Conclusions: We suggest that Pitx2 is involved in the cardiac outflow tract septation by promoting and/or maintaining the number and the remodeling process of the mesoderm progenitor cells. Pitx2 influences the expression of transcription factors and signaling molecules involved in the differentiation of the cushion mesenchyme during heart development.
Copyright © 2013 Wiley Periodicals, Inc.
Figures




Similar articles
-
The FGF-BMP signaling axis regulates outflow tract valve primordium formation by promoting cushion neural crest cell differentiation.Circ Res. 2010 Nov 12;107(10):1209-19. doi: 10.1161/CIRCRESAHA.110.225318. Epub 2010 Sep 16. Circ Res. 2010. PMID: 20847311 Free PMC article.
-
BMP2 expression in the endocardial lineage is required for AV endocardial cushion maturation and remodeling.Dev Biol. 2017 Oct 1;430(1):113-128. doi: 10.1016/j.ydbio.2017.08.008. Epub 2017 Aug 6. Dev Biol. 2017. PMID: 28790014 Free PMC article.
-
Foxp1 regulates cardiac outflow tract, endocardial cushion morphogenesis and myocyte proliferation and maturation.Development. 2004 Sep;131(18):4477-87. doi: 10.1242/dev.01287. Development. 2004. PMID: 15342473
-
Molecular regulation of atrioventricular valvuloseptal morphogenesis.Circ Res. 1995 Jul;77(1):1-6. doi: 10.1161/01.res.77.1.1. Circ Res. 1995. PMID: 7788867 Review.
-
Nfatc1 directs the endocardial progenitor cells to make heart valve primordium.Trends Cardiovasc Med. 2013 Nov;23(8):294-300. doi: 10.1016/j.tcm.2013.04.003. Epub 2013 May 10. Trends Cardiovasc Med. 2013. PMID: 23669445 Free PMC article. Review.
Cited by
-
Pitx genes in development and disease.Cell Mol Life Sci. 2021 Jun;78(11):4921-4938. doi: 10.1007/s00018-021-03833-7. Epub 2021 Apr 12. Cell Mol Life Sci. 2021. PMID: 33844046 Free PMC article. Review.
-
A cellular atlas of Pitx2-dependent cardiac development.Development. 2019 Jun 14;146(12):dev180398. doi: 10.1242/dev.180398. Development. 2019. PMID: 31201182 Free PMC article.
-
Current Perspectives in Cardiac Laterality.J Cardiovasc Dev Dis. 2016 Dec 9;3(4):34. doi: 10.3390/jcdd3040034. J Cardiovasc Dev Dis. 2016. PMID: 29367577 Free PMC article. Review.
-
Intrauterine Programming of Diabetes Induced Cardiac Embryopathy.Diabetes Obes Int J. 2019;4(3):202. Epub 2019 May 6. Diabetes Obes Int J. 2019. PMID: 32537569 Free PMC article.
-
Pitx2c orchestrates embryonic axis extension via mesendodermal cell migration.Elife. 2018 Jun 28;7:e34880. doi: 10.7554/eLife.34880. Elife. 2018. PMID: 29952749 Free PMC article.
References
-
- Abu-Issa R, Smyth G, Smoak I, Yamamura K, Meyers EN. Fgf8 is required for pharyngeal arch and cardiovascular development in the mouse. Development. 2002;129:4613–4625. - PubMed
-
- Amendt BA, Sutherland LB, Semina EV, Russo AF. The molecular basis of Rieger syndrome. Analysis of Pitx2 homeodomain protein activities. J Biol Chem. 1998;273:20066–20072. - PubMed
-
- Arima Y, Miyagawa-Tomita S, Maeda K, Asai R, Seya D, Minoux M, Rijli FM, Nishiyama K, Kim KS, Uchijima Y, Ogawa H, Kurihara Y, Kurihara H. Preotic neural crest cells contribute to coronary artery smooth muscle involving endothelin signalling. Nat Commun. 2012;3:1267. - PubMed
-
- Brown CB, Boyer AS, Runyan RB, Barnett JV. Antibodies to the Type II TGFbeta receptor block cell activation and migration during atrioventricular cushion transformation in the heart. Dev Biol. 1996;174:248–257. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

