Aftins increase amyloid-β42, lower amyloid-β38, and do not alter amyloid-β40 extracellular production in vitro: toward a chemical model of Alzheimer's disease?
- PMID: 23364140
- PMCID: PMC5039020
- DOI: 10.3233/JAD-121777
Aftins increase amyloid-β42, lower amyloid-β38, and do not alter amyloid-β40 extracellular production in vitro: toward a chemical model of Alzheimer's disease?
Abstract
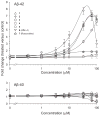
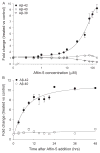
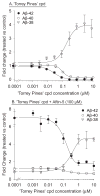
Increased production of amyloid-β (Aβ)42 peptide, derived from the amyloid-β protein precursor, and its subsequent aggregation into oligomers and plaques constitutes a hallmark of Alzheimer's disease (AD). We here report on a family of low molecular weight molecules, the Aftins (Amyloid-β Forty-Two Inducers), which, in cultured cells, dramatically affect the production of extracellular/secreted amyloid peptides. Aftins trigger β-secretase inhibitor and γ-secretase inhibitors (GSIs) sensitive, robust upregulation of Aβ42, and parallel down-regulation of Aβ38, while Aβ40 levels remain stable. In contrast, intracellular levels of these amyloids appear to remain stable. In terms of their effects on Aβ38/Aβ40/Aβ42 relative abundance, Aftins act opposite to γ-secretase modulators (GSMs). Aβ42 upregulation induced by Aftin-5 is unlikely to originate from reduced proteolytic degradation or diminished autophagy. Aftin-5 has little effects on mitochondrial functional parameters (swelling, transmembrane potential loss, cytochrome c release, oxygen consumption) but reversibly alters the ultrastructure of mitochondria. Aftins thus alter the Aβ levels in a fashion similar to that described in the brain of AD patients. Aftins therefore constitute new pharmacological tools to investigate this essential aspect of AD, in cell cultures, allowing (1) the detection of inhibitors of Aftin induced action (potential 'anti-AD compounds', including GSIs and GSMs) but also (2) the identification, in the human chemical exposome, of compounds that, like Aftins, might trigger sustained Aβ42 production and Aβ38 down-regulation (potential 'pro-AD compounds').
Figures
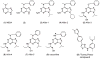


Similar articles
-
Induction of Amyloid-β42 Production by Fipronil and Other Pyrazole Insecticides.J Alzheimers Dis. 2018;62(4):1663-1681. doi: 10.3233/JAD-170875. J Alzheimers Dis. 2018. PMID: 29504531 Free PMC article.
-
Brain toxicity and inflammation induced in vivo in mice by the amyloid-β forty-two inducer aftin-4, a roscovitine derivative.J Alzheimers Dis. 2015;44(2):507-24. doi: 10.3233/JAD-140711. J Alzheimers Dis. 2015. PMID: 25298201
-
Independent generation of Abeta42 and Abeta38 peptide species by gamma-secretase.J Biol Chem. 2008 Jun 20;283(25):17049-54. doi: 10.1074/jbc.M802912200. Epub 2008 Apr 21. J Biol Chem. 2008. PMID: 18426795
-
γ-Secretase modulator in Alzheimer's disease: shifting the end.J Alzheimers Dis. 2012;31(4):685-96. doi: 10.3233/JAD-2012-120751. J Alzheimers Dis. 2012. PMID: 22710916 Review.
-
Oligomer Formation by Amyloid-β42 in a Membrane-Mimicking Environment in Alzheimer's Disease.Molecules. 2022 Dec 12;27(24):8804. doi: 10.3390/molecules27248804. Molecules. 2022. PMID: 36557940 Free PMC article. Review.
Cited by
-
Structural Analysis of the Simultaneous Activation and Inhibition of γ-Secretase Activity in the Development of Drugs for Alzheimer's Disease.Pharmaceutics. 2021 Apr 8;13(4):514. doi: 10.3390/pharmaceutics13040514. Pharmaceutics. 2021. PMID: 33917979 Free PMC article.
-
Prohibitin ligands: a growing armamentarium to tackle cancers, osteoporosis, inflammatory, cardiac and neurological diseases.Cell Mol Life Sci. 2020 Sep;77(18):3525-3546. doi: 10.1007/s00018-020-03475-1. Epub 2020 Feb 15. Cell Mol Life Sci. 2020. PMID: 32062751 Free PMC article. Review.
-
A comparison of cognitive decline in aged mice and mice treated with aftin-4.Sci Rep. 2024 Nov 16;14(1):28320. doi: 10.1038/s41598-024-79792-3. Sci Rep. 2024. PMID: 39550500 Free PMC article.
-
Decrease in p3-Alcβ37 and p3-Alcβ40, products of Alcadein β generated by γ-secretase cleavages, in aged monkeys and patients with Alzheimer's disease.Alzheimers Dement (N Y). 2019 Nov 7;5:740-750. doi: 10.1016/j.trci.2019.09.015. eCollection 2019. Alzheimers Dement (N Y). 2019. PMID: 31754625 Free PMC article.
-
An in vitro workflow of neuron-laden agarose-laminin hydrogel for studying small molecule-induced amyloidogenic condition.PLoS One. 2022 Aug 26;17(8):e0273458. doi: 10.1371/journal.pone.0273458. eCollection 2022. PLoS One. 2022. PMID: 36026506 Free PMC article.
References
-
- Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science. 2002;297:353–356. Erratum in: Science 297, 2209. - PubMed
-
- Benilova I, Karran E, De Strooper B. The toxic Aβ oligomer and Alzheimer’s disease: An emperor in need of clothes. Nat Neurosci. 2012;15:349–357. - PubMed
-
- Karran E, Mercken M, De Strooper B. The amyloid cascade hypothesis for Alzheimer’s disease: An appraisal for the development of therapeutics. Nat Rev Drug Discov. 2011;10:698–712. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

