Oxidative folding in the mitochondrial intermembrane space in human health and disease
- PMID: 23364613
- PMCID: PMC3588022
- DOI: 10.3390/ijms14022916
Oxidative folding in the mitochondrial intermembrane space in human health and disease
Abstract
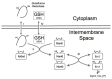
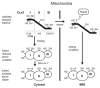
Oxidative folding in the mitochondrial intermembrane space (IMS) is a key cellular event associated with the folding and import of a large and still undetermined number of proteins. This process is catalyzed by an oxidoreductase, Mia40 that is able to recognize substrates with apparently little or no homology. Following substrate oxidation, Mia40 is reduced and must be reoxidized by Erv1/Alr1 that consequently transfers the electrons to the mitochondrial respiratory chain. Although our understanding of the physiological relevance of this process is still limited, an increasing number of pathologies are being associated with the impairment of this pathway; especially because oxidative folding is fundamental for several of the proteins involved in defense against oxidative stress. Here we review these aspects and discuss recent findings suggesting that oxidative folding in the IMS is modulated by the redox state of the cell.
Figures
Similar articles
-
The MIA pathway: a key regulator of mitochondrial oxidative protein folding and biogenesis.Acc Chem Res. 2015 Aug 18;48(8):2191-9. doi: 10.1021/acs.accounts.5b00150. Epub 2015 Jul 27. Acc Chem Res. 2015. PMID: 26214018 Free PMC article.
-
Oxidation of Arabidopsis thaliana COX19 Using the Combined Action of ERV1 and Glutathione.Antioxidants (Basel). 2023 Nov 1;12(11):1949. doi: 10.3390/antiox12111949. Antioxidants (Basel). 2023. PMID: 38001802 Free PMC article.
-
Erv1 of Arabidopsis thaliana can directly oxidize mitochondrial intermembrane space proteins in the absence of redox-active Mia40.BMC Biol. 2017 Nov 8;15(1):106. doi: 10.1186/s12915-017-0445-8. BMC Biol. 2017. PMID: 29117860 Free PMC article.
-
The mitochondrial intermembrane space: a hub for oxidative folding linked to protein biogenesis.Antioxid Redox Signal. 2013 Jul 1;19(1):54-62. doi: 10.1089/ars.2012.4855. Epub 2012 Oct 3. Antioxid Redox Signal. 2013. PMID: 22901034 Review.
-
The Erv1-Mia40 disulfide relay system in the intermembrane space of mitochondria.Biochim Biophys Acta. 2008 Apr;1783(4):601-9. doi: 10.1016/j.bbamcr.2007.12.005. Epub 2007 Dec 15. Biochim Biophys Acta. 2008. PMID: 18179776 Review.
Cited by
-
Glutathionylated and Fe-S cluster containing hMIA40 (CHCHD4) regulates ROS and mitochondrial complex III and IV activities of the electron transport chain.Redox Biol. 2020 Oct;37:101725. doi: 10.1016/j.redox.2020.101725. Epub 2020 Sep 15. Redox Biol. 2020. PMID: 32971361 Free PMC article.
-
CHCHD4 Oxidoreductase Activity: A Comprehensive Analysis of the Molecular, Functional, and Structural Properties of Its Redox-Regulated Substrates.Molecules. 2025 May 10;30(10):2117. doi: 10.3390/molecules30102117. Molecules. 2025. PMID: 40430290 Free PMC article. Review.
-
Disulfide driven folding for a conditionally disordered protein.Sci Rep. 2017 Dec 5;7(1):16994. doi: 10.1038/s41598-017-17259-4. Sci Rep. 2017. PMID: 29208936 Free PMC article.
References
-
- Schmidt O., Pfanner N., Meisinger C. Mitochondrial protein import: From proteomics to functional mechanisms. Nat. Rev. Mol. Cell. Biol. 2010;11:655–667. - PubMed
-
- Baker K.P., Schatz G. Mitochondrial proteins essential for viability mediate protein import into yeast mitochondria. Nature. 1991;349:205–208. - PubMed
-
- Dessi P., Whelan J. Temporal regulation of in vitro import of precursor proteins into tobacco mitochondria. FEBS. Lett. 1997;415:173–178. - PubMed
-
- Dudley P., Wood C.K., Pratt J.R., Moore A.L. Developmental regulation of the plant mitochondrial matrix located Hsp70 chaperone and its role in protein import. FEBS Lett. 1997;417:321–324. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

