Radical scavenging capacity of methanolic Phillyrea latifolia L. extract: anthocyanin and phenolic acids composition of fruits
- PMID: 23364751
- PMCID: PMC6270162
- DOI: 10.3390/molecules18021798
Radical scavenging capacity of methanolic Phillyrea latifolia L. extract: anthocyanin and phenolic acids composition of fruits
Abstract
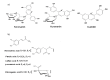
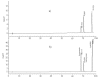
Radical scavenging capacity of a crude methanolic extract from the fruits of Phillyrea latifolia L., commonly known as green olive tree or mock privet, was investigated with reference to anthocyanin standards, as flavonoids, and phenolic acid standards, as phenylpropanoids. Characterization with high performance liquid chromatography-diode array detection (HPLC-DAD) indicated the presence of keracyanin, kuromanin, cyanidin, ferulic acid, caffeic acid and rosmarinic acid at amounts of 289.1, 90.4, 191.4, 225.2, 221.2 and 190.1 mg/100 g fresh weight (FW) of fruits, respectively. Chlorogenic and p-coumaric acids were found to exist in lower amounts. Trolox equivalent antioxidant capacity (TEAC) and IC(50) values of the plant extract were found to be 1.8 mM Trolox equivalents (TE)/g FW of fruits and 69.4 µg/mL, respectively, indicating the close radical scavenging activity of the extract to those of keracyanin and p-coumaric acid. The crude methanolic P. latifolia L. fruit extract was seen to be fairly potent in radical scavenging. Total phenolic content (TPC) of the plant extract was found to be 1652.9 mg gallic acid equivalent (GAE)/100 g FW of fruits.
Figures

References
-
- Hung H.C., Joshipura K.J., Jiang R., Hu F.B., Hunter D., Smith-Warner S.A., Colditz G.A., Rosner B., Spiegelman D., Willett W.C. Fruit and vegetable intake and risk of major chronic disease. J. Natl. Cancer Inst. 2004;92:1577–1584. - PubMed
-
- Harborne J.B. Nature, Distribution and Function of Plant Flavonoids. In: Cody B., Middleton E., Harborne J.B., editors. Plant Flavonoids Biology and Medicine. Alan Liss; New York, NY, USA: 1986. pp. 15–24. - PubMed
-
- Shahidi F., Wanasundara P.K.J. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr. 1992;32:67–103. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

