Optimizing the 6-min walk test as a measure of exercise capacity in COPD
- PMID: 23364913
- PMCID: PMC3515028
- DOI: 10.1378/chest.11-2702
Optimizing the 6-min walk test as a measure of exercise capacity in COPD
Abstract
Background: It is uncertain whether the effort and expense of performing a second walk for the 6-min walk test improves test performance. Hence, we attempted to quantify the improvement in 6-min walk distance if an additional walk were to be performed.
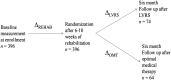
Methods: We studied patients consecutively enrolled into the National Emphysema Treatment Trial who prior to randomization and after 6 to 10 weeks of pulmonary rehabilitation performed two 6-min walks on consecutive days (N = 396). Patients also performed two 6-min walks at 6-month follow-up after randomization to lung volume reduction surgery (n = 74) or optimal medical therapy (n = 64). We compared change in the first walk distance to change in the second, average-of-two, and best-of-two walk distances.
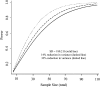
Results: Compared with the change in the first walk distance, change in the average-of-two and best-of-two walk distances had better validity and precision. Specifically, 6 months after randomization to lung volume reduction surgery, changes in the average-of-two (r = 0.66 vs r = 0.58, P = .01) and best-of-two walk distances (r = 0.67 vs r = 0.58, P = .04) better correlated with the change in maximal exercise capacity (ie, better validity). Additionally, the variance of change was 14% to 25% less for the average-of-two walk distances and 14% to 33% less for the best-of-two walk distances than the variance of change in the single walk distance, indicating better precision.
Conclusions: Adding a second walk to the 6-min walk test significantly improves its performance in measuring response to a therapeutic intervention, improves the validity of COPD clinical trials, and would result in a 14% to 33% reduction in sample size requirements. Hence, it should be strongly considered by clinicians and researchers as an outcome measure for therapeutic interventions in patients with COPD.
Figures

Similar articles
-
Relation of exercise capacity with lung volumes before and after 6-minute walk test in subjects with COPD.Respir Care. 2014 Nov;59(11):1687-95. doi: 10.4187/respcare.03082. Epub 2014 Sep 23. Respir Care. 2014. PMID: 25249649
-
Reliability, repeatability and sensitivity to change of externally and self-paced walking tests in COPD patients.Respir Med. 2003 Apr;97(4):407-14. doi: 10.1053/rmed.2002.1462. Respir Med. 2003. PMID: 12693802
-
The endurance shuttle walking test: a responsive measure in pulmonary rehabilitation for COPD patients.Chron Respir Dis. 2006;3(1):3-9. doi: 10.1191/1479972306cd077oa. Chron Respir Dis. 2006. PMID: 16509172
-
Heart rate recovery after the 6-min walk test is related to 6-min walk distance and percutaneous oxygen saturation recovery in patients with COPD.Respirology. 2015 May;20(4):671-3. doi: 10.1111/resp.12510. Epub 2015 Mar 20. Respirology. 2015. PMID: 25800043
-
Effects of inspiratory muscle thixotropy on the 6-min walk distance in COPD.Respir Med. 2008 Jul;102(7):970-7. doi: 10.1016/j.rmed.2008.02.007. Epub 2008 Apr 18. Respir Med. 2008. PMID: 18396028 Clinical Trial.
Cited by
-
Comparison between effects of pressure support and pressure-controlled ventilation on lung and diaphragmatic damage in experimental emphysema.Intensive Care Med Exp. 2016 Dec;4(1):35. doi: 10.1186/s40635-016-0107-0. Epub 2016 Oct 19. Intensive Care Med Exp. 2016. PMID: 27761886 Free PMC article.
-
Role of Acupuncture in the Treatment of COPD: An Overview of Systematic Reviews.Int J Gen Med. 2021 Mar 25;14:1079-1092. doi: 10.2147/IJGM.S300270. eCollection 2021. Int J Gen Med. 2021. PMID: 33790637 Free PMC article. Review.
-
Use of positive expiratory pressure during six minute walk test: results in patients with moderate to severe chronic obstructive pulmonary disease.Multidiscip Respir Med. 2013 Mar 14;8(1):19. doi: 10.1186/2049-6958-8-19. Multidiscip Respir Med. 2013. PMID: 23497658 Free PMC article.
-
The effectiveness and safety of Chuna manual therapy adjuvant to Western medicine in patients with chronic obstructive pulmonary disease: Study protocol for a randomized, single-blind, investigator-initiated, pilot trial.Medicine (Baltimore). 2021 Sep 24;100(38):e27217. doi: 10.1097/MD.0000000000027217. Medicine (Baltimore). 2021. PMID: 34559113 Free PMC article.
-
Lung volume reduction surgery for diffuse emphysema.Cochrane Database Syst Rev. 2016 Oct 14;10(10):CD001001. doi: 10.1002/14651858.CD001001.pub3. Cochrane Database Syst Rev. 2016. PMID: 27739074 Free PMC article.
References
-
- Salzman SH. The 6-min walk test: clinical and research role, technique, coding, and reimbursement. Chest. 2009;135(5):1345-1352 - PubMed
-
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166(1):111-117 - PubMed
-
- National Emphysema Treatment Trial Research Group A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348(21):2059-2073 - PubMed
-
- Patel SA, Sciurba FC. Emerging concepts in outcome assessment for COPD clinical trials. Semin Respir Crit Care Med. 2005;26(2):253-262 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- N01HR76113/HR/NHLBI NIH HHS/United States
- N01HR76104/HR/NHLBI NIH HHS/United States
- N01HR76105/HR/NHLBI NIH HHS/United States
- N01 HR076101/HL/NHLBI NIH HHS/United States
- N01HR76108/HR/NHLBI NIH HHS/United States
- N01HR76117/HR/NHLBI NIH HHS/United States
- N01HR76109/HR/NHLBI NIH HHS/United States
- N01HR76116/HR/NHLBI NIH HHS/United States
- N01HR76112/HR/NHLBI NIH HHS/United States
- N01HR76107/HR/NHLBI NIH HHS/United States
- N01HR76102/HR/NHLBI NIH HHS/United States
- N01HR76118/HR/NHLBI NIH HHS/United States
- N01HR76115/HR/NHLBI NIH HHS/United States
- N01HR76111/HR/NHLBI NIH HHS/United States
- N01HR76114/HR/NHLBI NIH HHS/United States
- N01HR76103/HR/NHLBI NIH HHS/United States
- P50HL084948/HL/NHLBI NIH HHS/United States
- N01HR76110/HR/NHLBI NIH HHS/United States
- P50 HL084948/HL/NHLBI NIH HHS/United States
- N01 HR076119/HL/NHLBI NIH HHS/United States
- N01HR76106/HR/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

